Journal de Pharmacie Clinique
MENUFeedback from a university hospital one year after the “simplified” reform of early and expanded access programm: a growing complexity for health professionals Volume 41, issue 4, Décembre 2022
- Key words: early and expanded access, pharmaceuticals, health services accessibility/legislation and jurisprudence, innovation
- DOI : 10.1684/jpc.2022.0492
- Page(s) : 149-57
- Published in: 2022
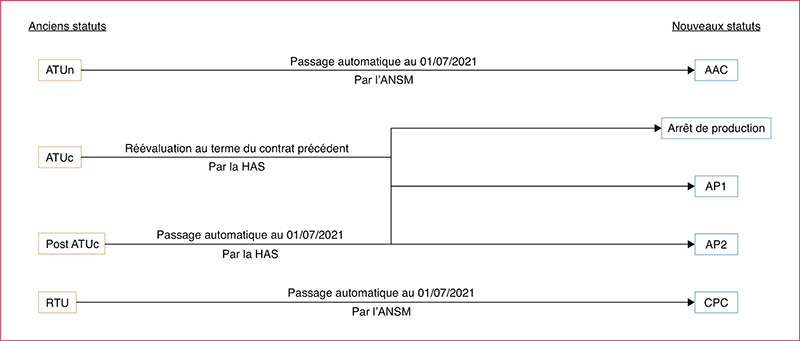
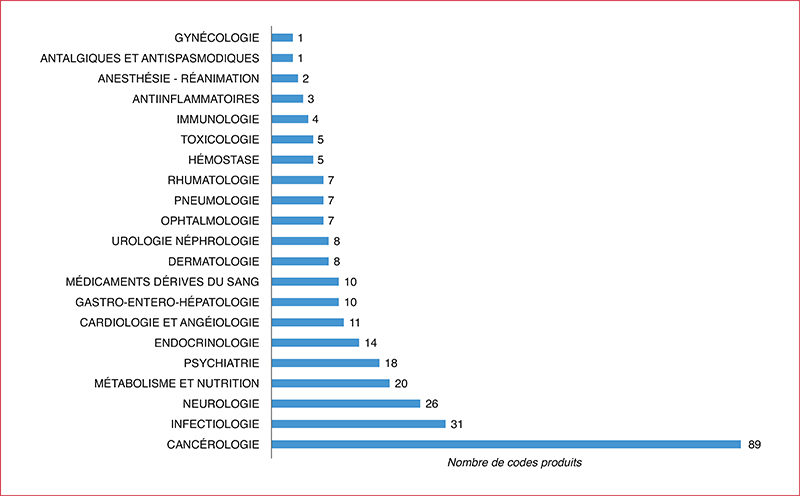
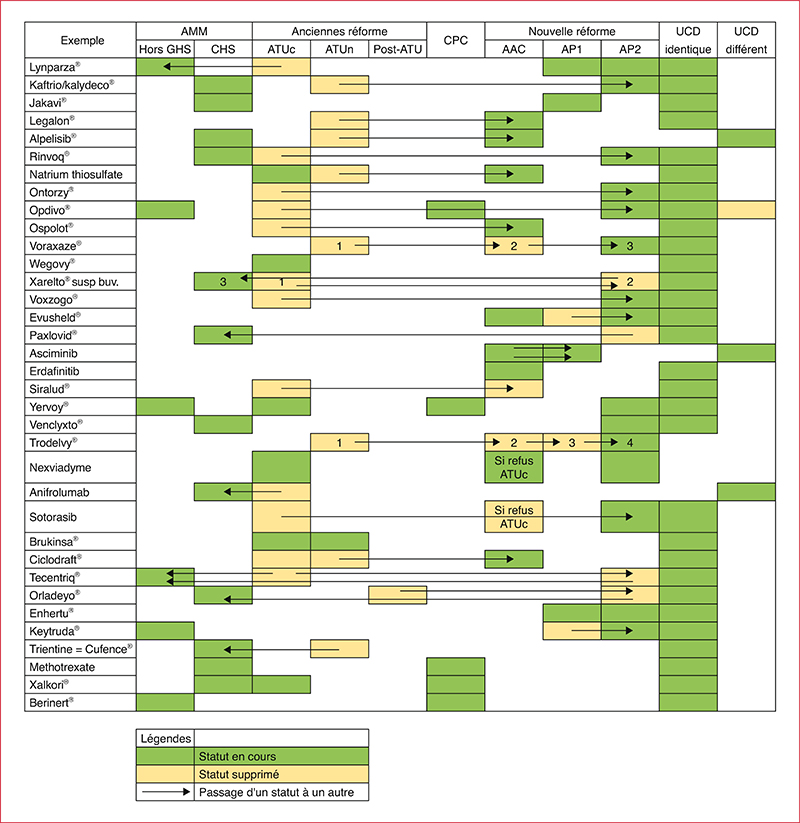
The reform of early and expanded access programm (EAP) starting on the 1st July 2021 is intended to simplify access for patients to innovative treatments. However, it has led to organisational rearrangements within health establishments and it is particularly complex due to many possible circuits of request and supply. An assessment of the situation between the 1st July 2021 and the 1st July 2022 was carried out in our hospital to evaluate the impact of the reform on the organisation and the pharmaceutical time dedicated to this activity. The derogatory status impact 198 molecules (51% compassionate access authorization, 31% early access authorization, 12% compassionate acess framework and 6% cohort temporary authorization). Access authorisations are specific to each laboratory or indication (40% connection to a computing platform required for data collection and/or orders), making access to treatment more complex. Their management mainly involves three sectors of the pharmacy: cancer chemotherapy, retrocessions and the procurement of drugs. One third of the references concern multi-status drugs for which the duplication of GEF codes is sometimes essential but not without risk (billing error, stock management, indication coding). The amount of pharmaceutical time required varies from case to case, but requires multidisciplinary consultation in order to consider the problems of management, the supply circuit, financial monitoring and IT configuration. Some one hundred changes in status have been identified and increase the time required on hospital pharmacists for monitoring, follow-up and communication with physicians for medicines in EAP.