Hépato-Gastro & Oncologie Digestive
MENUAdverse effects of roton pump inhibitor's: Myths and realities Volume 28, issue 8, Octobre 2021
- Key words: inhibitors of proton pump, adverse effects, good use
- DOI : 10.1684/hpg.2021.2223
- Page(s) : 974-85
- Published in: 2021
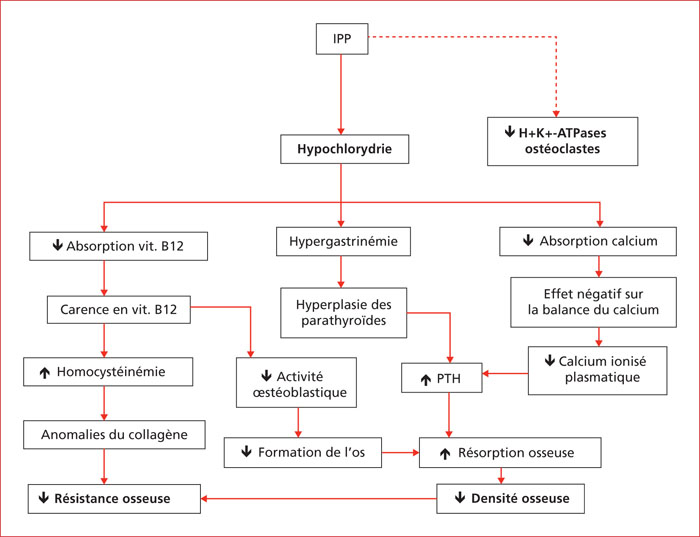
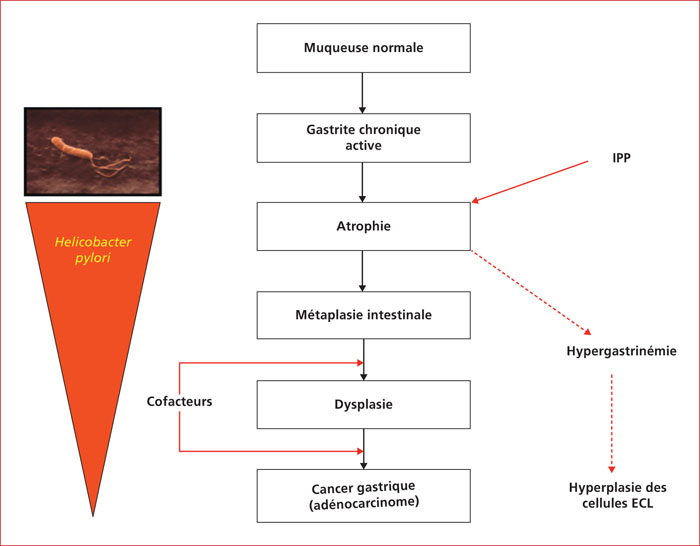
Proton pump inhibitor's (PPI) are one of the most prescribed therapeutic classes. The respect of the indications is at the center of a public health issue, the rates of inappropriate prescription ranging from 25 to 70% according to the data in the literature. The problem of security in the use of PPIs has recently attracted the attention of the media and lay people. Gastroenterologists are frequently asked about the suitability and safety of treatment with PPI. In order to avoid untimely and inappropriate discontinuation of an indicated treatment, prescribers should objectively assess the evidence to support a possible cause-and-effect relationship between an adverse reaction and taking PPIs. Despite the long list of potential side effects of PPIs reported in numerous publications in recent years, the quality of the evidence provided in the literature is often insufficient to establish a causal relationship between treatment with PPIs and many of the effects unwanted reported. Thus, many reported side effects could be the result of methodological biases observed in observational studies, often retrospective. The correct use of PPIs is the first step in preventing side effects; it includes compliance with the indications, adjusting the dosage to the minimum effective dose, reconsidering the treatment with each new prescription and is part of our medical responsibility to limit the occurrence of adverse effects. The knowledge of these effects and their risk of occurrence can help prevent, detect and treat them early appropriately. In the event of a proven indication for treatment with PPI, there is currently no sufficient scientific evidence to impose treatment to be discontinued in order to limit the occurrence and intensity of potential side effects. The aim of this work is to review the main side effects attributed to PPIs reported in the literature.