Hématologie
MENUThe genomic landscape of adult B-cell precursor acute lymphoblastic leukaemia Ahead of print
- Key words: B-cell precursor acute lymphoblastic leukaemia (BCP-ALL), oncogenesis, genetic alterations, next-generation sequencing
- DOI : 10.1684/hma.2020.1518
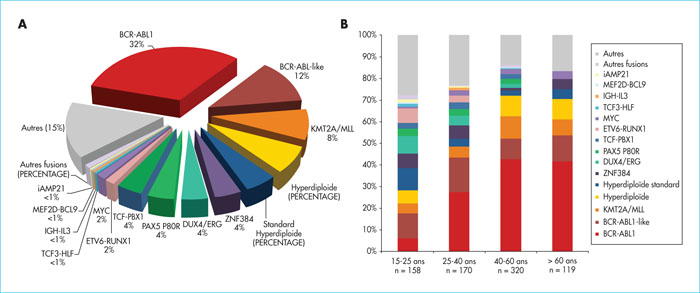
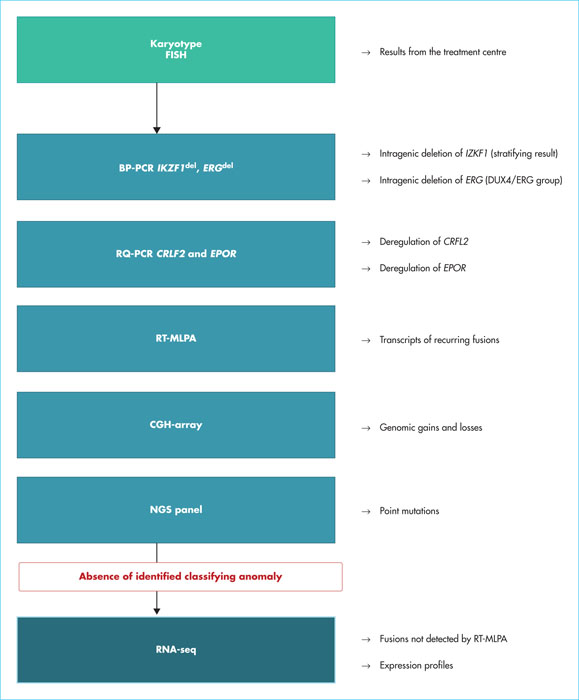
Adult B-cell precursor acute lymphoblastic leukaemia (BCP-ALL) is usually classified based on the presence or absence of Philadelphia chromosome (Ph), although Ph- BCP-ALL is not a homogenous group in terms of genetic and clinical characteristics. In contrast to the long-standing role of cytogenetic classification in risk prediction and treatment stratification for paediatric BCP-ALL, genetic alterations and their prognostic relevance are poorly known in adult BCP-ALL. Recently, the development of high-throughput genome-wide analyses has allowed many recurrent alterations to be identified in children and adults with BCP-ALL. Ten years ago, the BCR-ABL1-like or Ph-like group was described in paediatric studies and the importance of this group in adults was subsequently recognised. Evaluation of new therapeutic options in this high-risk group is currently an important field in treatment development. More recently, genomic alterations have been identified, defining novel subtypes, such as DUX4/ERG, ZNF384, ME2D and PAX5P80R. These multiple “classifying” alterations, together with many secondary alterations, have created a new genomic landscape for adult BCP-ALL of unexpected complexity. Understanding the biology of these leukaemias and analysing their clinical features and response to drugs are rapidly evolving fields that should speed up the development of more precise and effective treatments.