Epileptic Disorders
MENUThe response to ACTH is determined early in the treatment of infantile spasms Volume 17, issue 1, March 2015
Infantile spasms (IS) are seizures that are frequently associated with poor developmental outcome. With treatment, an average of 16% of children with IS may experience a favourable outcome (Frost and Hrachovy, 2003). Even those patients with less favourable outcomes can benefit from treatment with variable degrees of developmental progress. Early and effective treatment may allow for improved developmental outcome (Riikonen, 2001; Eisermann et al., 2003; Kivity et al., 2004; Darke et al., 2010).
Although successful treatment of IS with adrenocorticotropic hormone (ACTH) was first described 55 years ago (Sorel and Dusaucy-Bauloye, 1958), the optimal ACTH dose and duration has not been determined. The 2004 American Academy of Neurology and the Child Neurology Society practice parameter could not recommend an ACTH dose or duration for IS due to lack of evidence (Mackay et al., 2004). The 2012 update of this practice parameter suggested that low-dose (e.g. 20 IU/day) ACTH may be as effective as high-dose (150 IU/m2/day) ACTH for the short-term treatment of IS (Go et al., 2012). However, high-dose ACTH regimens remain in widespread use based on studies reporting high rates of electroclinical remission (Baram et al., 1996). An optimal treatment duration has not been determined for either low-dose or high-dose regimens.
The 2010 United States consensus report on IS advocates for changes in treatment if early electroclinical remission is not achieved (Pellock et al., 2010). Understanding when remission typically occurs with ACTH will help to inform decisions about early treatment changes among non-responders. When we established our IS management protocol in 2012, we changed our ACTH regimen to a four-week short course, to be consistent with the Food and Drug Administration-approved package insert for Acthar gel. This transition in regimen created a unique opportunity to compare outcomes between the adopted four-week short course and the previously used 12-week long course. We hypothesized that successful ACTH treatment is determined within two weeks of initiation, regardless of the duration of treatment. The aim of this study was to characterize the time from ACTH initiation to clinical remission among a cohort of ACTH responders.
Methods
We conducted a retrospective chart review of all IS patients managed at our institution from January 2009 to September 2013. Subjects were identified using an EEG database (search terms: “hypsarrhythmia” and “infantile spasms”) and hospital discharge codes (345.60, 345.61). Patients who did not receive ACTH were excluded. Two high-dose natural ACTH gel protocols were used during the study period; a 12-week regimen used prior to September 2012 published by Snead and colleagues (Snead et al., 1983) and a four-week regimen used after September 2012 published by Baram and colleagues (Baram et al., 1996). The 12-week ACTH regimen was dosed as follows: 75 IU/m2 twice daily the first week, 75 IU/m2 daily the second week, 75 IU/m2 every other day the third and fourth week, and this was followed by a slow eight-week taper. The four-week ACTH regimen included two weeks at 75 IU/m2 twice daily, followed by a rapid taper over two weeks.
We defined ACTH response as complete remission of clinical IS sustained for greater than or equal to 28 days beginning at any time within the ACTH course and remission of hypsarrhythmia (or definite EEG improvement if hypsarrhythmia was absent at baseline). A relapse was assigned if IS resumed after the initial remission. For ACTH responders, the diagnostic and post-ACTH EEG tracings were reviewed by a paediatric epileptologist (JRM) to assess for electrographic improvement.
The Mann-Whitney U, Pearson chi-square and Fisher exact tests were used to compare continuous and categorical variables. The log-rank test was used to compare time to remission between ACTH protocols. The one-way ANOVA was used to compare the time to remission between our cohort and two previously published cohorts. All statistical analyses were performed using SPSS Version 21 (SPSS Inc., Chicago, IL, USA). Significance was set at the 5% level. This study was approved by the Internal Review Board at Nationwide Children's Hospital.
Results
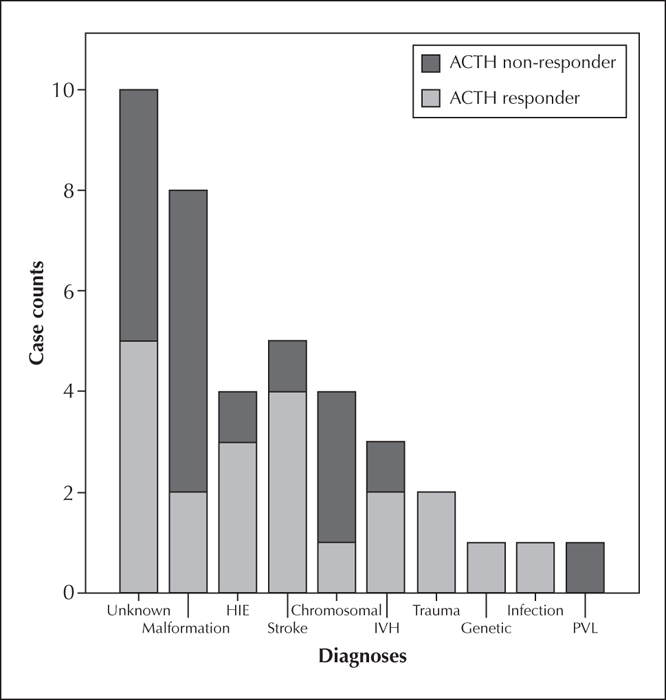
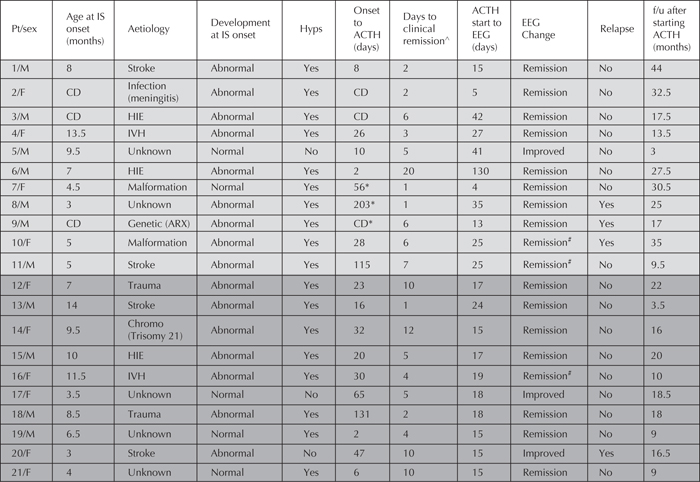
Of 87 IS patients identified, 42 were treated with ACTH. One patient was excluded from analysis because the response to ACTH could not be determined and two patients were excluded because no post-ACTH EEG was performed. Of the remaining 39 patients, 21 responded to ACTH (54%). Table 1shows ACTH responders by treatment protocol, demographics and outcome. There was no significant difference in response between those receiving a long course (11/25, 48%) and a short course (10/14, 71%) of ACTH (p=0.15). Aside from the dates of treatment and the duration of treatment, there were no significant differences between patients in the long course and short course groups including: age at diagnosis (p=0.39), sex (p=0.43), age at ACTH initiation (p=0.87), and the number of patients with less than or equal to 30 days between IS onset and starting ACTH (p=0.34). Twenty-six percent of patients (10/39; seven in the long course group and three in the short course group) had unknown aetiology. There was no significant difference (p=0.78) in ACTH response when comparing patients with unknown aetiology to those with a documented aetiology. Figure 1 shows similar ACTH response, regardless of aetiology. We identified one serious adverse event in the long course group. After failing to respond to ACTH, a patient with trisomy 21 died of sepsis eight days after the dose of ACTH was increased back to the initial dose of 150 U/m2/day, 20 days into the treatment course. There were no serious side effects in the short course group.
The date of IS onset could be determined in 32 patients. For these patients, the median interval from IS onset to the start of ACTH (treatment lag) was 29 days (mean: 43.6 days; range: 2-203; interquartile range [IR]: 9.25-63). The treatment lag did not significantly differ based on ACTH response (responders: median of 24.5 days, range of 3-95, IR of 9.5-58; non-responders: median of 27 days, range of 3-95, IR of 8-79; p=0.72) or protocol (long course: median of 28 days, range of 2-203, IR of 9-83; short course: median of 31 days, range of 2-131, IR of 13.5-59; p=0.92).
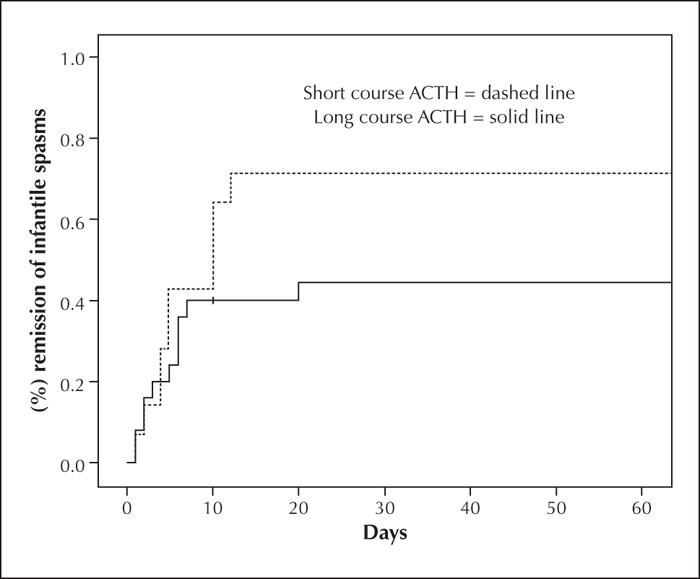
The mean time to clinical remission (first seizure-free day) for the 21 responders was 5.8 days (median: 5 days; range: 1-20). Figure 2 illustrates the percent of seizure remission as a function of ACTH treatment days; time to remission did not significantly differ between the long course and short course groups (p=0.51). The patient with clinical remission at 20 days did so only after a second course of high-dose ACTH (75 IU/m2 twice daily) was started 19 days into treatment. Among responders, this was the only example of a protocol deviation identified. A relapse of IS after an initial remission was uncommon in both protocols (three in the long course group and one in the short course group).
Among ACTH responders, the median interval from ACTH initiation to the post-ACTH EEG was 18 days (mean: 25.5 days; range: 4-130; IR: 15-26); this interval was not significantly different between the long course (median: 25 days; range: 4-130; IR: 13-41) and short course (median: 17 days; range: 15-24; IR: 15-18) groups (p=0.25). For ACTH responders, the median duration from ACTH initiation to last follow-up was 25 months (mean: 23 months; range: 3-44) in the long course group and 16.3 months (mean: 14.3 months; range: 3.5-22) in the short course group (table 1).
Discussion
Our study of IS patients is the first to compare the number of days to clinical remission in a group of consecutive ACTH responders treated with either a short course or long course of ACTH. In our cohort, response was determined within two weeks of starting ACTH in 96% and within three weeks in 100%. Additional treatment with ACTH beyond three weeks (for up to 12 weeks) did not yield additional responders. Although not powered for this analysis, there was no significant difference in the response rates for those treated with a long course (44%) or a short course (71%) of ACTH.
We identified two other studies that reported the time to remission in cohorts that received the same ACTH regimens used in our study. In the study by Snead and colleagues (utilizing the same high-dose, long course ACTH regimen used in our study), the mean time to clinical remission was 5.9 days in 14 responders (Snead et al., 1989). In the study by Baram and colleagues (utilizing the same high-dose, short course regimen used in our study), the mean time to clinical remission was approximately 3.2 days in 13 responders (Baram et al., 1996). Our mean time to clinical remission of 5.8 days matches the 5.9 days reported by Snead and colleagues, and is not statistically different from the 3.2 days reported by Baram and colleagues (p=0.15). If we combine our mean response time with the response times from these two prior studies, the mean time to clinical remission is about five days, with the response to ACTH determined within two weeks in 94% (44/47) of responders and within three weeks in 100%.
In their study, Snead and colleagues had two patients with clinical remission beyond two weeks of starting ACTH (one at 15 days and one at 21 days) (Snead et al., 1989). One of our patients experienced clinical remission 20 days after ACTH initiation. While cause-and-effect is not proven, this patient's ACTH was increased back to 75 IU/m2 twice daily, 19 days into the original treatment protocol. Despite this protocol deviation, we included this patient in our analysis as a long course ACTH responder. However, our current treatment protocol does not include the reintroduction of high-dose ACTH following an initial treatment failure due to limited available safety data.
One argument for the use of longer ACTH protocols has been the possible reduction in the rate of relapse. The number of relapses in our study was small (three in the long course group and one in the short course group). Although the low relapse rate following the short course of ACTH is somewhat reassuring, our small study size and the brief duration of follow-up in two patients (one due to death and another lost to follow-up) prevents us from drawing conclusions about relapse and the duration of ACTH treatment.
While the rate of electroclinical remission was not the primary outcome measure, the rate of electroclinical remission in this study was well within the range reported for IS clinical trials (42-87%; Pellock et al., 2010) and nearly identical to the rate reported in the Class I study by Hrachovy and colleagues (1994). Our inclusion of all IS cases could have biased our results, with a lower overall remission rate compared to some prospectively designed clinical trials where the most severely affected patients might be excluded. However, our electroclinical remission rate may appropriately represent clinical practice.
In addition to the retrospective study design and small study population, our study included other limitations. Given that our ACTH cohorts were treated in two different time periods, we cannot exclude the possibility that other confounders affected our results. Our study also does not address the pharmacodynamic differences between natural and synthetic ACTH or the differences between high-dose and low-dose ACTH. For example, differences in potency may affect the timing of response. In addition, our EEG review was not blinded to the duration of ACTH treatment and three post-ACTH EEGs did not include sleep. We acknowledge the importance of sleep during the EEG, given that findings to suggest an IS-associated epileptic encephalopathy may be present only during sleep (Watanabe et al., 1993). The clinical response and the absence of any subsequent signs of IS in these patients over a mean of 18 months (range: 10-35) of follow-up provides some reassurance that they experienced a remission without relapse. Although our current protocol includes a post-treatment overnight EEG to confirm electroclinical remission, this was not done consistently in this cohort. The absence of prolonged EEG monitoring to confirm electroclinical remission in ACTH responders is a shortcoming of this study. Of the responders who had a post-ACTH EEG, all but one had the EEG within 42 days (table 1); this outlier EEG was performed four months after starting ACTH, raising the possibility of spontaneous remission (the rate of which is estimated to be 7% at four months) (Hrachovy et al., 1991).
This study provides Class IV evidence that among IS patients, the response to ACTH is determined early in the treatment course for most patients. Given the importance of rapid remission (Darke et al., 2010), clinicians should consider adding or changing treatment if IS do not resolve within two weeks of ACTH initiation. While we acknowledge that an IS remission might occur after three weeks in some patients, the low likelihood of a late response may not justify the potential for adverse events with long-duration ACTH treatment. Further study is needed to determine the optimal ACTH regimen for IS.
Acknowledgements and disclosures
This research was funded by an internal grant from the Nationwide Children's Hospital Research Institute. We thank Tim P. Held for his assistance in the search of our EEG database.
This work was presented at the 2013 Annual Meeting of the American Epilepsy Society in poster format.
The authors have no conflicts of interest to declare.