Epileptic Disorders
MENUEpileptic ictal strabismus: a case report and review of the literature Volume 20, issue 4, August 2018
Ictal strabismus, sometimes associated with epileptic nystagmus, is an extremely rare epileptic phenomenon, suggestive of cortical involvement in monocular eye movement control (Galimberti et al., 1998; Thurtell et al., 2009; Schulz et al., 2013). We describe a patient with ictal disconjugate contraversive horizontal eye deviation of cortical origin as the main clinical feature of a focal seizure. We also review the extant literature on this intriguing ictal phenomenon.
Case study
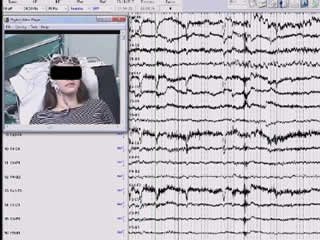
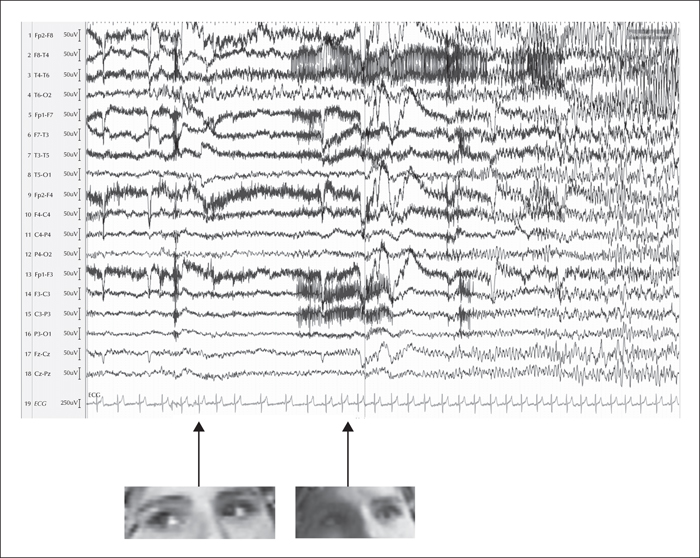
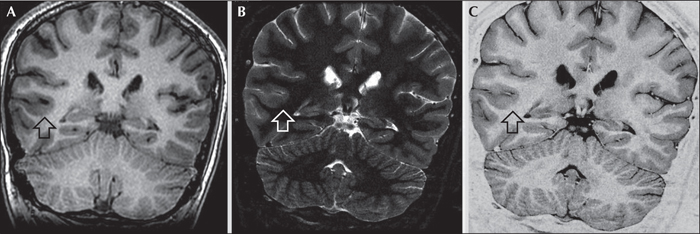
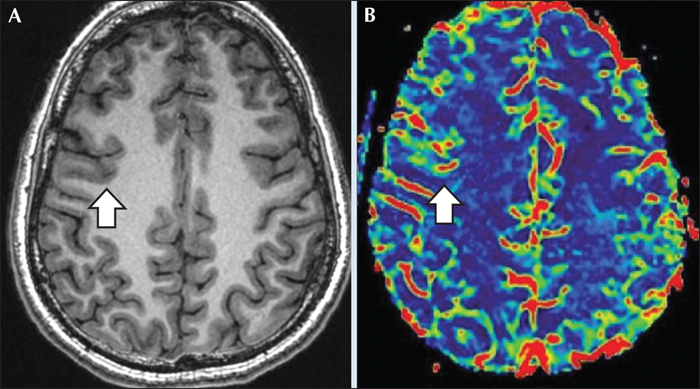
A 17-year-old, previously healthy, right-handed woman was referred to our centre for diagnostic evaluation. Four days earlier, at around 7.00 a.m., the sister, who was sleeping in a nearby room, had heard the patient crying and when entering the patient́s room she had found her unable to react, with foaming of the mouth. The patient was brought to the emergency department, where brain MRI and routine blood examinations were performed yielding normal results. During video-EEG recording, performed during the following days, the patient had a seizure characterized by initial rightward conjugate deviation of the eyes, followed by convergent strabismus due to adduction of the right eye towards the nose without conjugate left eye abduction (esotropia) (figure 1, video sequence). When the strabismus appeared, the patient blinked. Tachycardia was also detected immediately after the blink. Strabismus was not accompanied by nystagmus. Afterwards, she did not react to external stimulation, had leftward deviation (version) of the head, and then a bilateral tonic-clonic seizure. Postictal and interictal status were unremarkable; more specifically, neuro-ophthalmological examination revealed neither nystagmus nor altered eye motility. After the seizure, she did not report having experienced any double vision or visual disturbance. Ictal EEG showed a rhythmic theta activity over the right posterior temporal region (related to rightward conjugate deviation of the eyes), spreading to ipsilateral fronto-central regions when strabismus appeared. Treatment with levetiracetam up to 1,000 mg/day was started. Two months after the seizure described above, the patient experienced a further epileptic seizure which was unwitnessed. The dosage of levetiracetam was therefore increased to 1,500 mg/day without further seizures (overall length of follow-up was 10 months). 3.0 Tesla MRI was performed peri-ictally, and images were acquired with a 3.0 Tesla MRI scanner (Signa Architect, GE Healthcare, Milwaukee, WI). The protocol included a 3D MPR T1-weighted sequence (BRAVO), a 3D FLAIR (CUBE) sequence, a thin-slice (3.5-mm) axial DWI sequence, a thin-slice axial (3-mm) FSE T2-weighted sequence, coronal T2 and inversion recovery (IR) thin-section (1-mm) images, as well as a dynamic susceptibility contrast (DSC) MR perfusion sequence and a 3D MPR T1-weighted sequence after contrast media administration. 3.0 Tesla MRI showed a focal area of slightly thickened cortex and blurred cortical-subcortical junction in the posterior section of the right temporal lobe, suggestive of cortical dysplasia (figure 2). Perfusion-weighted imaging showed focal cortical-subcortical cerebral blood flow increase in the right frontal eye field area, correlating with the ictal epileptic activity (figure 3).
Discussion
We describe a patient with ictal strabismus due to adduction of the right eye without conjugate left eye abduction (esotropia) as the main clinical feature of a focal seizure. Ictal strabismus is an epileptic phenomenon which has been very rarely reported in the medical literature.
Intermittent esotropia associated with attacks of dizziness has been interpreted as an equivalent of absences in a three-year girl, with an EEG showing generalized symmetric and synchronous 3/s spike-wave discharges; squinting resolved completely after therapy with ethosuximide (Gusek-Schneider et al., 2000).
Other reports have described strabismus as a clinical expression of focal seizures.
Tischler and colleagues described a nine-year-old girl with strabismus due to in-turning of the right eye and jerk nystagmus on lateral gaze with preserved consciousness, associated with spike discharges in the left parietal-occipital region (Tischler et al., 1996). Authors attributed this clinical phenomenon to epileptic activation of a smooth pursuit pathway.
Galimberti and co-workers described a patient with epileptic skew deviation, diplopia, and oscillopsia associated with a right-beating nystagmus (Galimberti et al., 1998). The EEG recording showed epileptic activity over left parieto-occipital regions. The authors suggested that these ocular motor signs were “derived from an ictal activation of the vestibular cortex, which in turn activated descending projections to the vestibular nuclei, leading to both a dynamic (right-beating nystagmus) and a static (skew deviation) vestibular imbalance” (Galimberti et al., 1998).
Thurtell and colleagues described two epilepsy patients with ictal strabismus occurring during electrical stimulation of the frontal eye fields or during focal seizures spreading from the supero-posterior Sylvian bank to the ipsilateral frontal lobe (Thurtell et al., 2009). As in the patient we describe, the interictal neuro-ophthalmological findings were normal, and neuroimaging did not show any abnormality in the brainstem. Hence, the authors concluded that the cortical frontal eye field plays a role in both contralateral version and vergence eye movements. In this regards, Rasmussen and Penfield had already observed that stimulation of the precentral gyrus immediately anterior to the central sulcus (Area 8) resulted in contralateral eye deviation (Rasmussen and Penfield, 1948), although less often it could also result in convergence (Smith, 1944). Subsequently, Jampel (1960) reported that stimulation of the frontal lobe of the macaque brain could elicit vergence eye movements, and a further animal study has demonstrated that the frontal eye field is a cortical area involved in the generation of disconjugate eye movements (Ferraina et al., 2000).
Subsequently, Schulz and co-workers described a patient with ictal nystagmus affecting only one eye, associated with ictal diplopia; symptoms disappeared after resection of right frontal focal cortical dysplasia (Schulz et al., 2013). Whereas this finding could support the hypothesis of an exclusively cortical involvement in monocular eye movement control, as previously suggested (Thurtell et al., 2009), the presence of monocular nystagmus and spasm of the contralateral eye observed on interictal neuro-ophthalmological examination “argue for an irregular activation of both the cortical frontal eye field and the brainstem” (Schulz et al., 2013).
Our patient had an initial rightward conjugate eye deviation associated with a right posterior temporal discharge, followed by ictal disconjugate contraversive (leftward) horizontal eye deviation with convergent strabismus resulting from adduction of the right eye without conjugate left eye abduction. It was difficult to ascertain the presence of impaired awareness at the very onset of ictal strabismus in our patient. However, when the strabismus appeared, she blinked, as if she was initially aware of and disturbed by diplopia (but when questioned postictally, the patient did not recall any symptom of double vision occurring immediately before loss of consciousness). Immediately after the appearance of strabismus, she was not able to react to external stimuli. The ictal tachycardia, starting immediately after the blink, could simply be the consequence of emotional stress and heightened adrenergic drive, but may also represent a symptom of early involvement of the temporal and orbito-frontal cortex at seizure onset (Stefanidou et al., 2015).
No baseline alterations in eye motility or brainstem abnormality were found. It is therefore reasonable to conclude that in our patient, the ictal strabismus had an exclusive cortical origin.
The initial rightward conjugate eye deviation associated with right posterior temporal rhythmic theta activity (ipsiversive eye deviation) is likely to be the clinical manifestation of a seizure originating from the inferio-posterior temporal lobe (seizure onset zone) (Zhang et al., 2017). MRI confirmed the presence of an epileptogenic lesion (Lüders et al., 2006) in that region, showing cortical dysplasia in the posterior section of the right temporal lobe. Due to the low spatial resolution of scalp EEG, we could not identify with precision the symptomatogenic zone underlying ictal strabismus. Ictal activity was not recorded invasively, hence it is impossible to draw definitive conclusions regarding possible propagation of epileptic activity from the right inferio-posterior temporal to ipsilateral frontal lobe. It is possible that the epileptic activity remained initially confined to the right temporal lobe and cortical regions nearby. If so, cortical regions involved in controlling versive and vergence eye movements, such as the parietal eye field and dorsolateral prefrontal cortex (Coubard and Kapoula, 2006), or the involvement of middle temporal and medial superior temporal areas, which control normal smooth pursuit movement (Tijssen et al., 1992), may have contributed to the ictal strabismus.
However, the patient eventually had a leftward head version, indicating epileptic activation of the frontal eye field (inferior/medial frontal gyrus) (Bonelli et al., 2007; Noachtar and Arnold, 2008). Hence, the ictal strabismus might have been the clinical expression of epileptic activity involving the right frontal eye field (symptomatogenic zone) (Thurtell et al., 2009; Schulz et al., 2013), as further supported by the concomitant appearance of rhythmic theta activity over the right fronto-central region and by the perfusion sequences showing focal cortical-subcortical cerebral blood flow increase in the right frontal eye field area, correlating with the ictal epileptic activity.
Acknowledgements and disclosures
We are grateful to Andrea Orioli for graphical support.
No funding was received related to the preparation of this article. Francesco Brigo has received speakers’ honoraria from Eisai and PeerVoice, payment for consultancy from Eisai, and travel support from Eisai, ITALFARMACO, and UCB Pharma. The other co-authors have no conflict of interest to declare.