Médecine de la Reproduction
MENUBiological consequences of advanced maternal age in reproduction Volume 21, issue 3, Juillet-Août-Septembre 2019
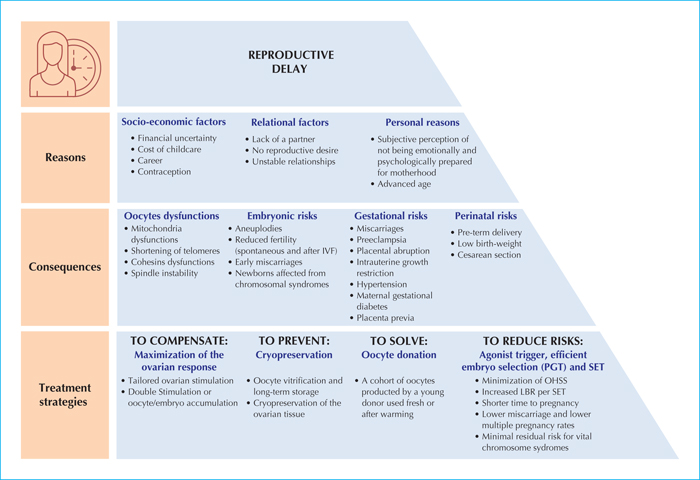
The close association between increasing maternal age and decreasing success in conceiving is becoming alarmingly relevant in view of the worldwide trend in postponing pregnancy. As a matter of fact, especially in Western societies, childbearing has been progressively delayed also due to the change in women's perception of themselves in the society. Several socio-economic, relational and personal reasons are at the root of the reproductive delay. The increasing educational background, the desire for career and improved social conditions, as well as the misleading perception that infertility might be solved through medical treatments are the major reasons behind this trend (figure 1). IVF success is strongly associated with maternal age and 35 years represent the lower threshold to define a woman of advanced maternal age (AMA) [1]. AMA patients are subjected to a sustained increasing rate of embryonic aneuploidies and it is estimated that, in women over 43, the probability of producing an euploid blastocyst is lower than 5% [2]. The risk for embryonic aneuploidies is mainly related with the physiological and age-dependent depletion of the ovarian reserve and the reduction of the oocyte/embryo competence, defined as the ability of an embryo to result in a live birth. In the oocyte, the main mechanisms impaired by ageing are related with energy production and chromosome segregation during meiosis. Before embryonic genome activation (EGA) no transcription occurs, therefore, only maternal information inherited from the oocyte holds the control of the first and crucial steps of embryogenesis [3, 4]. In addition, in order to effectively sustain the initial embryo development and reach EGA correctly, a synchronous oocyte nuclear and cytoplasmic maturation is crucial. The oocyte genetic stability could be jeopardized by ageing during the quiescence which goes from fetal development to menarche, a period in which the meiosis is physiologically arrested in prophase I. In particular, the longer this period of quiescence the greater the risk of damage. Human prophase of meiosis I is characterized by a particular state of association between the four sister chromatids held together by at least one DNA crossing-over. This physical attachment, known as tetrad, that allows for alignment recombination and segregation of the homologous chromosomes, must be maintained for years. In AMA women, oocytes are subjected to a further prolonged arrest period resulting in a weakening of the bonding structure. The consequent formation of univalent or sister chromatids splitting are high incidence events correlated with ageing, through a still unclear mechanism. During maternal oogenesis, the premature separation of sister chromatids (PSSC) in meiosis I is the most common segregation error [5]. Less frequent, but still relevant, meiosis I or meiosis II non-disjunction events cause maternal meiotic impairments [6]. For all these reasons, as well, in AMA patients, the infertility work-up and treatment are recommended already after just 6 months of regular unprotected intercourse.
A comprehensive diagnostic picture of each patient's reproductive health requires, beyond the assessment of the ovarian reserve markers (FSH, AMH and antral follicle count), also the definition of thyroid functioning, coagulation disorders, infections, sperm quality, tubal patency, and uterine pathologies. Similarly, a proper counseling which must cover any gestational complication like hypertension, preeclampsia, diabetes, placental abruption, intrauterine growth restriction, placenta previa, low birth-weight, pre-term delivery and fetal deaths, is also pivotal.
Currently, there are no therapies to counteract the decline and/or confer competence to a gamete/embryo. Clinicians and embryologists can only try to outline the most suitable strategy to maximize the ovarian response, obtain the ideal number of oocytes for each woman, and safeguard their residual competence during any kind of manipulation in vitro.
The next paragraphs are focused on the putative oocyte molecular and cellular mechanisms affected by ageing that lead to a reduced oocyte/embryo competence and on the therapeutic approaches to treat AMA women. Both the current established strategies and the emerging approaches are treated.
Biological causes for age-related oocyte/embryo decreased competence
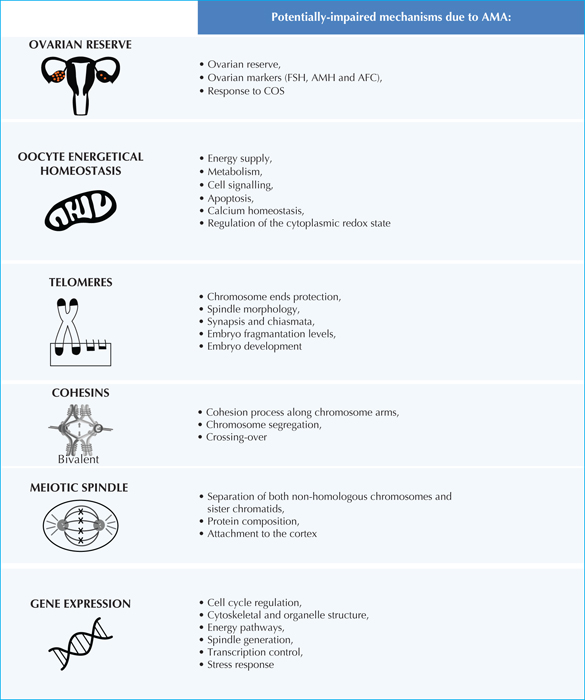
Mitochondria
The mitochondria, as powerhouse of the cell, play a crucial role in the process of aging. They produce most of the cells’ energy supply. The energy for transcription and translation during oocyte maturation, fertilization, and embryonic development is provided by these organelles that convert chemical energy from nutrients into adenosine triphosphate (ATP). Mitochondria are also involved in metabolism, cell signaling, apoptosis, calcium homeostasis and regulation of the cytoplasmic red-ox state (figure 2). They are characterized by their own genome (mtDNA), which replicates independently from cellular division. The mutation rate of mtDNA is 25-times higher than genomic DNA, due to a less efficient DNA repair mechanism, and the longer the quiescence period the heavier its damage. After fertilization, the mitochondria of the sperm are immediately degraded, except for rare reported cases of mtDNA heteroplasmy [7], and new mitochondria in preimplantation embryos are generated only at the blastocyst stage. Of note, structural mitochondria issues (swelling, vacuolization and cristae alteration), as well as impaired metabolic activity and progressive alteration of the membrane activity potential characterize aged oocytes [8]. Moreover, it has been speculated that poor-quality mitochondria may lead to a precocious arrest in embryo development in AMA women [9]. Lower mtDNA concentration and more deletions have been reported from polar bodies (PBs) [10] and granulosa cells (GCs) [11]. For all these reasons, embryo development, quality and implantation might be dependent on the mitochondria within the oocytes and older patients show significantly decreased concentration of the mtDNA in their oocytes [12].
Several clinical and basic science investigations across the last years hypothesized a correlation between mtDNA content and fertilization, embryo viability or reproductive competence. Fragouli and colleagues were the first to claim that mtDNA levels from a trophectoderm biopsy might associate with the implantation potential of euploid blastocysts [13]. In this regard, they conducted a blinded prospective non-selection study involving 199 blastocysts where elevated levels of mtDNA were shown in euploid blastocysts failing to result into an ongoing pregnancy. However, several other studies published to date highlighted highly discordant results, absence of a proper validation and of a biological rationale to support a clinical value for mtDNA analysis from trophectoderm biopsies. Therefore, this is just an intriguing hypothesis which has not been reliably demonstrated [14].
Another group preferred testing the spent culture media at the cleavage stage for its content in mtDNA and reported a significant association between its quantitation in day3 of preimplantation development in vitro and embryo developmental potential to blastocyst [15]. The data produced to date are interesting and benefit from the great advantage of non-invasiveness in the sample collection procedure. Certainly, this workflow requires further investigation and validation from future studies, conducted also in different settings and from different operators.
Beyond molecular testing, some authors instead suggested an experimental therapeutic strategy: autologous mitochondria isolated from egg precursor cells present in the ovarian cortical tissue might be transferred into aged oocytes to improve their developmental/reproductive potential, according to an approach named Autologous Germline Mitochondrial Energy Transfer (AUGMENT) [16]. However, also AUGMENT betrayed its promises when finally tested in a recent triple-blind, single-center, randomized, controlled pilot study conducted by Labarta and colleagues [17]. In fact, the study was interrupted because AUGMENT not only did not improve any IVF outcome, but also decreased the blastulation rate of the treated oocytes.
Telomeres
Chromosome ends are protected from deterioration by the telomeres and their associated proteins. These structures are specialized regions of repetitive nucleotide sequences that act as a conservative cap, converting the blunt ends of linear chromosomes to closed loops, thus preventing DNA damage. During each replication in dividing cells, telomeres are subjected to a progressive shortening. When telomeres become critically short and inefficient, the unprotected chromosome ends set off genomic instability, cell cycle arrest and cell death. With advancing paternal age sperm telomeres lengthen; on the contrary, oocytes’ telomeres are among the shortest ones in the human body. Telomere attrition in the oocytes leads to meiotic dysfunction, spindle morphological alterations, reduced synapsis and chiasmata, embryo high fragmentation levels, delayed development or developmental arrest. In the male germline, the length of telomeres is preserved (figure 2). In fact, throughout a man's life-span, spermatogonia show telomerase activity, the reverse transcriptase capable of restoring telomere repeats lost with each round of DNA replication. Conversely, oocytes and cleavage stage embryos express low or unmeasurable levels of activity.
Telomeres’ attrition in oocytes begins during the fetal phase of oogenesis and lasts in aged ovary. The leading causes can be identified in oxidative stress, prolonged cell cycle arrest and in the reduced activity of the telomerase. It has been demonstrated that the telomeres are shorter in oocytes who experienced IVF failure or recurrent miscarriages [18], as well as in oocytes resulting in fragmented or aneuploid embryos [19]. Moreover, the telomerase becomes active in the embryo only between the late morula and the blastocyst stage.
Cohesins
The meiotic process during gametogenesis is a highly error-prone event that leads to the onset of chromosomal aneuploidies. The separation of homologous chromosomes and sister chromatids, in meiosis I and meiosis II respectively, is particularly prone to alterations. For this reason, the proper structure and organization of chromosomes during the meiotic process is crucial for the production of euploid gametes. The cohesins are part of a family of proteins strictly involved in the cohesion process along chromosome arms. They keep the bivalents intact in MI oocytes and hold together the centromere of sister chromatids in MII ones. Two distinct cohesin complexes are present during the meiotic process: a pericentric cohesin complex, required for the segregation of the chromosomes during the nuclear divisions, and a chromosomal arm complex, involved in the crossing-over (figure 2). Genetic recombination during the crossing-over is not equally distributed in the genome. For instance, in the pericentric regions, it occurs less frequently [20]. A correct and efficient organization of the cohesin complex in the pericentric region is pivotal to prevent the formation of pericentric meiotic double-stand breaks and crossovers. Dysfunctions in cohesins cluster during MI and MII oocytes stages may result in segregation errors. Several studies in mouse models demonstrated that the cohesin decay increases with oocyte ageing. Similarly, Tsutsumi [21] showed that in aged women, the high incidence of PSSC is often associated to cohesin imbalance. This is probably an effect of the reduced activity of all the regulatory proteins co-acting to prevent an anticipated removal of the cohesins with ageing.
Notably, a strict interaction of cohesions and telomeres has been reported in mice [22]. For this reason, it is assumed that the cohesins as well are affected by the same age-related issues that affect the telomeres. Cohesin dysfunctions, therefore, might be considered amongst the leading causes of maternal age-related aneuploidy.
Meiotic spindle
The spindle apparatus is a highly dynamic structure built on more than a hundred proteins that constitute the centrosome, a non-membrane-bound organelle required to separate both homologous chromosomes and sister chromatids during the meiotic nuclear division [23]. Unlike the centrosome of somatic cells, the one present in the oocyte is acentriolar. It is made up of proteins such as pericentrin, centrin, γ-tubulin and NuMa (Nuclear Mitotic Apparatus protein). Aberrations in the assembly of the spindle result into a high incidence of aneuploidies in AMA women. The integrity of this structure is maintained by a set of regulatory proteins including kinases (e.g., protein kinase A, MAPK, calmodulin kinase II) that, as reported by Qiao et al in 2014 [24], change in their compositions and impairment in their function lead to loss of spindle integrity. It was also hypothesised that an abnormal calcium elevation may result in premature cortical granule exocytosis and spindle detachment from the cortex determining the loss of signal transductions required to maintain spindle integrity. Moreover, the reduced amount of ATP resulting from a compromised mitochondrial activity in AMA women, concurs to a flawed spindle assembly. For all these reasons, young oocyte deeply differ from aged ones in both spindle morphology and orientation within the cell. Specifically, about 80% of the oocytes in AMA women show elongated or smaller spindle shape, as well as reduced microtubular foci in the cortex [23]. Lastly, also the composition of the “spindle assembly checkpoint” (SAC) is compromised in aged oocytes (figure 2). In fact, key SAC components, such as Bub1 protein, are progressively degraded as the oocytes age, causing a precocious loss of cohesion between sister chromosomes in the MI phase of meiosis [25].
Gene expression
Gene expression is another fundamental network of mechanisms potentially impaired in AMA oocytes. The expression of oocyte genes, in a variety of major functional categories implicated in cell cycle regulation, cytoskeletal structure and organelle preservation, energy pathways, spindle generation, transcription control, and stress responses, are influenced by maternal age (figure 2). These results are corroborated by complementary extensive studies in the murine model. In humans, it has been estimated that 5% of all transcripts in MII aged oocytes display modified amounts with respect to young women oocytes [26] also due to an altered pattern of epigenetic modifications [27].
Recently, much information has been accumulated also to support the relevance of circadian rhythms and clock-genes in an optimal reproductive performance. Some authors reported that specific molecular circadian clock-genes, existing in human luteinized granulosa cells, show a decreased expression in older women [28] and are probably essential for processes such as ovulation and steroidogenesis.
Current clinical strategies for the treatment of AMA patients
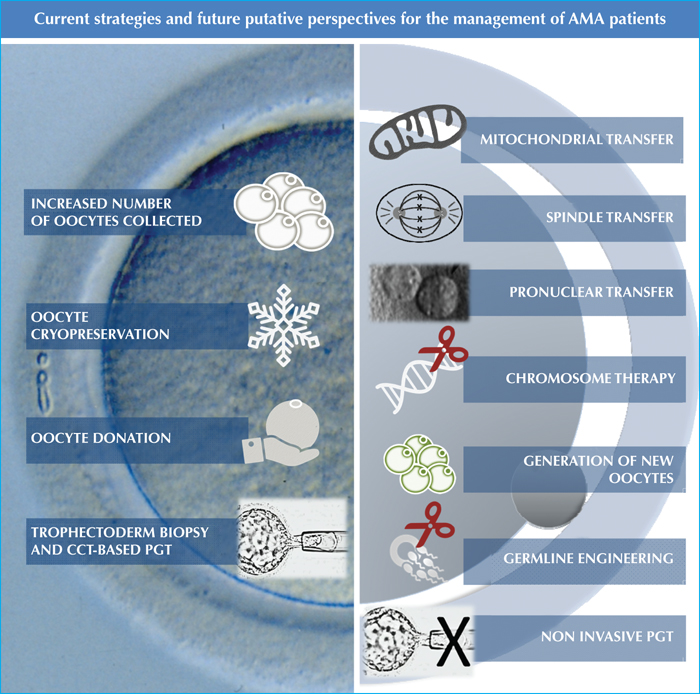
The management of AMA patients should be planned on the basis of both updated scientific evidences and couple-specific evaluations aimed at achieving the best attainable outcomes. A proper counseling is indeed pivotal to this end. The ideal workflow entails an accurate evaluation of the ovarian reserve and the personalization of the ovarian stimulation to collect an adequate number of oocytes in each woman (figure 3). Moreover, AMA women are also subject to increased risks of maternal mortality and morbidity compared to younger patients and therefore need special care to increase the efficiency of any IVF treatment while also ensuring its safety (reduce the risk for ovarian hyperstimulation syndrome [OHSS], miscarriage and multiple pregnancies). The strategy to fulfil both these premises encompass i) maximizing the ovarian response and adopt a freeze-all policy to minimize the risk of OHSS, and ii) enhancing embryo selection to identify reproductively competent embryos, thereby shortening the time to achieve a pregnancy, adopting a single embryo transfer policy and possibly reducing also the miscarriage rate (figure 3).
Maximization of the ovarian response
Maximizing the response to the ovarian stimulation in AMA patients is the most reasonable strategy to counteract the effect of aging and to compensate the reduced competence of the female gametes (figures 1, 3). The ideal number of oocytes to collect in order to achieve a pregnancy has been defined as ∼15 when accounting only fresh embryo transfers [29]. Conversely, when the total number of newborns is investigated with a cumulative perspective accounting both fresh and frozen transfers, the larger the number of oocytes the better the outcome [30]. Controlled ovarian stimulation (COS) can only support the growth of the recruited follicles, but it cannot create them. The aim is to collect the highest possible number of oocytes to obtain at minimum one competent embryo. The maximum threshold of gonadotrophins to this end is 300-375 IU/day for FSH and 75-150 IU/day for LH. Although LH is not always recommended, it can be useful in AMA as it counteracts the decreased sensitivity to exogenous FSH caused by aging, in the form of a decreased production of androgens [31]. OHSS is a iatrogenic complication which can occur when attempting to fully exploit the ovarian reserve with conventional/high dosage COS protocols. Although more frequent in young women, AMA patients with good ovarian reserve are at risk of OHSS. For this reason, “cycle segmentation” strategy is recommended to minimize its occurrence: COS and oocytes/embryos cryopreservation are conducted on a menstrual cycle, while vitrified-warmed embryo transfer is performed in a subsequent non-stimulated cycle on a physiological endometrium (figures 1, 3). Another COS approach available to limit OHSS is known as “mild stimulation”, since it is based on the administration of low doses of gonadotrophins. However this practice shows several disadvantages: the number of retrieved oocytes is limited and more stimulation cycles are required, the risk of no response to COS and cycle cancellation are higher, its cost-effectiveness is unclear especially in AMA patients who need to retrieve the highest possible number of oocytes in a short timeframe [32]. Also pharmacological co-treatments have been proposed to improve the IVF outcomes in AMA patients with reduced ovarian reserve, e.g. growth hormone [33], testosterone [34], dehydroepiandrosterone [35]. Nevertheless, their use require further studies, because the data are not concordant about an increased oocyte quantity/quality. The concept of tailoring ovarian stimulation protocols was therefore introduced. A thorough characterization of each patient is indeed pivotal in modern IVF. In this regard, poor responders to COS represent the group of patients more difficult to treat in IVF. A panel of experts known as the POSEIDON (Patient Oriented Strategies Encompassing Individualized Oocyte-Number) group introduced a new classification scheme for these patients, based on the oocyte yield after COS [36]. Specifically, poor prognosis patients are clustered into 4 groups: Group 1 (<35 years) and 2 (≥35 years) showing normal markers for ovarian reserve but sub-optimal response to gonadotrophins and representing the patients who can benefit from an increased dosage of FSH plus LH; Group 3 (<35 years) and 4 (≥35 years) who instead do not benefit from this strategy since they are characterized by a predicted low response already on the basis of their ovarian markers [37]. For Group 3 and 4, the most promising approaches are represented by oocyte/embryo accumulation in consecutive COS cycles [38] or double stimulation in the follicular and luteal phase of the same ovarian cycle (i.e. DuoStim) [39] (figure 1). Of note, the latter practice has been recently introduced after the demonstration that multiple waves of follicular recruitment arise during a single ovarian cycle [40], also in the luteal phase which is theoretically anovulatory. Despite the promising great results in the treatment of poor prognosis and oncological women, further investigations are required to define the safety and clinical efficiency of this novel unconventional COS protocol.
Oocyte cryopreservation
For years, embryo freezing has been considered the main strategy for fertility preservation, even if involving ethical, moral and legal concerns. In fact, slow-freezing of oocytes showed inconsistent efficacy and disappointing results until the introduction of vitrification. This rapid freezing protocol finally prevented the formation of ice crystals in the cell and led to an enormous improvement in the oocyte cryo-survival rates. In fact, at present, oocyte cryopreservation is considered the gold standard for fertility preservation for both medical (e.g. cancer, endometriosis) and non-medical reasons (social freezing) [41] (figures 1, 3). Oocyte vitrification is considered safe in terms of both survival rate and preservation of oocyte quality [42]: no increase in the aneuploidy rate [43], no modification of the gene expression profile [44], no decrease in the clinical and obstetrical outcomes [42]. However, two crucial variables can affect the efficacy of oocyte freezing procedures: maternal age at the time of cryopreservation and the number of stored oocytes. Specifically, based on detailed cost-effectiveness estimations, the maximum age limit for oocyte cryopreservation should be 37 yr [45]. Therefore, careful and clear information along with a proper counseling, should be provided to those patients willing to undergo oocyte cryopreservation. It is our duty to prevent false expectations for their future reproductive chances.
Oocyte donation
In AMA patients with a significant reduction of their ovarian reserve or recurrent IVF failures after several transfers of euploid embryos, the use of oocyte donation (OD) can be considered a valid alternative to adoption (figures 1, 3). In OD strategy, a cohort of fresh or cryopreserved oocytes is provided by a young donor. The pregnancy rate in women in their 50s is substantially high (>35%) demonstrating that the endometrium and the uterine apparatus maintain their function across the whole woman reproductive lifespan. It is reported that in 2014, up to 12% of IVF cycles in the USA were performed with donated oocytes. However, despite the high pregnancy rate, the patients undergoing such treatment must be guided toward a conscious decision based on the existing clinical evidence. It is crucial to inform the patients that ageing per se represents a risk factor for gestational diabetes, thrombophlebitis, proteinuria, premature rupture of the membranes, hemorrhage, pre-term birth and low birthweight, intrauterine growth restriction and abnormal placentation [46]. Furthermore, there are some ethical concerns about the transfer of a totally non-self embryo in a recipient. Some reviews and meta-analyses showed an increasing percentage of maternal complications and obstetric defects in OD pregnancies, compared to IVF cycles with own oocytes. Surprisingly, it was also highlighted that these risks are not related only to maternal age, but they arise also due to immune reactions especially impairing the placentation process and exposing AMA women to miscarriage and a maternal death incidence 2-4 times higher with respect to younger women [47].
Preimplantation genetic testing of aneuploidies
Safeguarding woman health and achieving a singleton pregnancy are goals of utmost importance, especially in AMA patients. In this regard, preimplantation genetic testing of aneuploidies (PGT-A) has been introduced to discriminate euploid embryos from aneuploid ones in a cohort produced during an IVF cycle, thereby preventing the transfer of the latter and their inherent reproductive consequences (implantation failure, miscarriage, conception of fetuses affected from chromosomal syndromes) (figures 1, 3). This strategy might lead to both a shorter time to reach a healthy pregnancy and a lower risk of miscarriage [48, 49]. Clearly, aneuploidy testing cannot confer competence to reproductively-incompetent embryos (i.e. the cumulative live birth rate per intention to treat is intrinsic to the embryos and the patients), but it represents the most efficient embryo selection scheme implemented to date in IVF to increase the live birth rate per transfer. When performed at the blastocyst stage with comprehensive chromosome testing techniques (CCT: array-CGH, SNP-array, quantitative-PCR or NGS), it allows the clinicians to confidently adopt a single euploid blastocyst transfer strategy rather than a double untested blastocyst transfer one, thereby also minimizing the risk of multiple gestations [50].
Across the last 30 years from the theorization of PGT, three different approaches have been developed and implemented clinically: i) Blastomere biopsy at the cleavage stage, whose clinical efficacy and efficiency were undermined by the issues of single cell analysis, its impact on embryo implantation potential and its initial inadequate association with 9-chomosome Fluorescence in-situ hybridization (FISH) technique [51-53]. All these reasons concurred to its unsuitability for clinical purposes ; ii) PB1 and PB2 analysis from oocytes/zygotes: an approach that has been reported not harmful for the resulting embryos and still more effective than untested ET, yet suffering from single cell analysis issues, a high workload for the laboratory and a diagnostic informativity limited to maternal meiotic errors, since paternal meiotic and mitotic missegregations cannot be detected [54, 55]; iii) Trophectoderm biopsy at the blastocyst stage: an approach that allows the retrieval of a multicellular biopsy from the section of the embryo that gives origin to the embryonic annexes, while keeping the inner cell mass (ICM) instead untouched. This last approach has been extensively reported safer, more informative and less time-consuming than the previous ones [51, 56].
Although PGT-A, when conducted at the blastocyst stage via CCT techniques, has been proven efficient in the hands of well-trained operators across several randomized controlled trials in reference laboratories [48, 49], chromosomal mosaicism still represents an important source of concerns. Chromosomal mosaicism is defined as the presence of cells with different karyotypes within the same embryo and it arises from mitotic missegregations after fertilization. While the co-existence of different lineages of aneuploid cells is not alarming, when both aneuploid and euploid cells are present in a blastocyst, the diagnostic reliability of PGT-A might result compromised due to an unavoidable sampling bias. Therefore, several studies have been conducted on human blastocysts donated to research in the last years. Specifically, these embryos were disaggregated in the ICM and several (up to 3) multicellular sections of the trophectoderm, which were then tested independently to confirm the reproducibility of their diagnoses: a design that allowed an estimation of a 5-10% prevalence of chromosomal mosaicism at the blastocyst stage [57]. This diagnostic limitation needs to be clearly acknowledged in the informed consent and thoroughly explained to the couples during counseling. Yet, if blastocyst stage PGT-A is aimed solely at the diagnosis of full chromosome meiotic aneuploidies [58], its clinical positive and negative predictive values (i.e. blastocyst diagnosed euploid that implant and blastocyst diagnosed aneuploid that do not implant) as high as about 50% and >95% [56], respectively, and its clinically-recognizable false negative error rate (i.e. aneuploid embryo diagnosed euploid and resulting in the conception of an affected fetus) is instead lower than 1% [59, 60]. Of note, such values are strictly dependent on the CCT technique adopted and might be even improved in the future. Until then, in case of the establishment of a pregnancy after PGT-A, non-invasive prenatal testing (NIPT) might be suggested as a confirmatory tool of PGT-A report.
A glimpse into the future strategies to treat AMA patients
The main social strategy to limit the increasing prevalence of infertility is prevention in the future generations. While lifestyle factors such as nutrition, exercise, smoking and drugs are now clearly recognized as causes of a reduced reproductive competence in the human gametes, the younger generations still underestimate the detrimental effect of aging per se. It is therefore crucial to encourage an abrupt change of mentality. At the same time, resources and efforts should be invested towards emerging fields of research. Many experimental strategies are currently under development to counteract the onset of infertility (figure 3).
Firstly, non-invasive embryo selection approaches to minimize gamete/embryo manipulation. For instance, the genomic, transcriptomic, miRNomic and proteomic analyses of by-products of IVF (e.g. follicular fluids after oocytes retrieval, cumulus cells after denudation, spent culture media after embryo culture in vitro) [61-63], as well as artificial intelligence adopted to analyze time-lapse videos of embryo preimplantation development [64], represent promising approaches for the future of ART. Especially the possibility of performing PGT on the spent culture media after IVF (non-invasive PGT) [65, 66] holds the potential for being revolutionary in IVF and therefore demands thorough and scrupulous validation. In particular the issues of exogenous, PB-derived and maternal DNA contamination and of limited embryonic DNA detectable in the media should be overcome to fine-tune this ground-breaking perspective.
Secondly, the definition of putative strategies to restore and/or safeguard the developmental competence of aged oocytes is a field that deserves attention [67]. The first strategy proposed with a therapeutic practice, called “cytoplasmic transfer”, which dates back to the early ‘90s and consists in transferring a small volume of donor's oocyte cytoplasm from a fertile woman to an infertile one [68] (figure 3). However, due to related ethical concerns, the U.S. Food and Drug Administration (FDA) banned this practice in 2002. As an alternative, autologous cytoplasm transfer from oogonial stem cells (OSCs) [16, 69], or from somatic cells of ovarian origin, such as granulosa or cumulus cells [70], has been proposed. Similarly, also spindle-chromosomal complex from a patient's mature oocyte to a young donor's ones has been investigated as a putative strategy [71]. Nevertheless, neither the safety nor the effectiveness of these methods have been clearly elucidated, and therefore these are still a fiction rather than a fact.
Thirdly, pioneering studies on “chromosomal therapy” have been recently conducted on both animal models and human cells. The purpose is to correct chromosomal impairments in germ cells or IVF embryos. For instance, the CRISPR/Cas9 system has been exploited by Zuo and colleagues [72] to successfully eliminating target trisomic chromosomes from embryos, murine cultured cells, in-vivo tissues, Induced Pluripotent Stem Cell (iPSCs) from a Down Syndrome individual and cancer cells ex vivo.
Lastly, the possibility of generating new gametes in vitro is extremely fascinating. In this regard, already in 2004 Johnson and colleagues reported the presence of mitotically-active OSCs in mouse, which are able to generate new gametes that can be fertilized [73] (figure 3). In 2018, OSCs have been isolated from human ovarian cortical fragments of menopausal and non-menopausal women by Silvestris and colleagues [74], who could demonstrate that these cells can differentiate and enter meiosis.
The future is certainly bright in the field of IVF, but all these ground-breaking practices must be further carefully studied to overcome the current limitations. Deeper insights, validations and ethical discussions are required before a clinical application might be foreseen for them.
Liens d’intérêt
Les auteurs déclarent n’avoir aucun lien d’intérêt en rapport avec cet article.
![]() This work is licensed under a
Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License
This work is licensed under a
Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License