Epileptic Disorders
MENUOne-year clinical experience of perampanel in Spain: a multicentre study of efficacy and tolerability Volume 18, issue 2, June 2016
- Key words: perampanel, adverse event, efficacy, AED treatment, epilepsy
- DOI : 10.1684/epd.2016.0824
- Page(s) : 173-80
- Published in: 2016
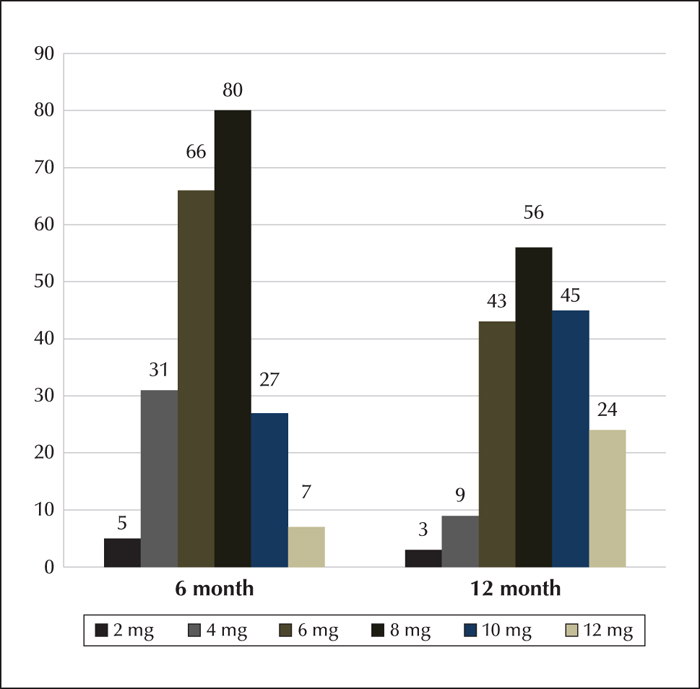
Perampanel, a non-competitive antagonist of the α-amino-3-hydroxy-5-methyl-4-isoxazole-propionic acid receptors, is the most recent antiepileptic drug available in Spain, marketed in January 2014. It was initially approved by the European Medicines Agency as adjunctive treatment for partial-onset seizures in patients 12 years and older, but recently also for primary generalized tonic-clonic seizures. Although clinical trials provide essential information about the drug, they do not reflect daily clinical practice. This retrospective study shows the initial experience with perampanel in 11 Spanish hospitals during its first year post-commercialisation. All patients who started perampanel treatment were included, but efficacy and tolerability were only assessed in those patients with a minimum follow-up period of six months. In total, 256 patients were treated with perampanel before September 2014, and 253 had an observational period of one year. After six months, 216/256 patients (84%) continued on perampanel and 180/253 (71.1%) completed one year of treatment. The mean number of previous antiepileptic drugs used was 6.83 and the median number of concomitant antiepileptic drugs was 2. The mean perampanel dose was 7.06 mg and 8.26 mg at six and 12 months, respectively. The responder rate was 39.5% and 35.9% at both follow-up points, respectively. Adverse events were experienced by 91/253 (35.5%) and resulted in withdrawal in 37 (14.6%). The most common adverse events were somnolence, dizziness, and irritability. We found no significant differences between concomitant use of enzyme-inducing and non-inducing antiepileptic drugs, regarding efficacy, adverse effects, or withdrawals. Irritability was not influenced by concomitant use of levetiracetam, relative to other drugs, but was more frequently observed in patients with a history of psychiatric problems or learning disabilities.