Epileptic Disorders
MENUEpilepsy surgery for polymicrogyria: a challenge to be undertaken Volume 20, issue 5, October 2018
Definition
The term polymicrogyria (PMG) refers to an excessive number of abnormally small, and partly fused, cortical gyri with shallow sulci and abnormal cortical lamination (Barkovich et al., 2005). MRI presents a characteristic gyral pattern with an irregular cortical surface, apparent cortical thickening, and a “stippled” grey-white junction (Leventer et al., 2010). PMG is one of the most common malformations of cortical development (MCD), accounting for approximately 20% of all MCDs (Leventer et al., 2010), and can be associated with other brain abnormalities, such as corpus callosum dysgenesis, cerebellar hypoplasia, schizencephaly (SCZ), periventricular and subcortical heterotopia, focal cortical dysplasia (FCD), and hemimegalencephaly (Wieck et al., 2005; Maillard et al., 2009; Ramantani et al., 2013a; Wang et al., 2016). PMG occurs due to disruptions of cerebral cortical development, most probably during the late stages of neuronal migration and early stages of cortical organization (Barkovich et al., 2005). Brain pathology shows abnormal development or loss of neurons in the middle and deep cortical layers, accompanied by an unlayered (two layers) or layered (four to six layers) cortical structure (Judkins et al., 2011). The underlying aetiology is presumably heterogeneous, including environmental as well as genetic factors. PMG occurs at the proximity of perinatal ischaemic insults (Barkovich et al., 1995), and may be linked to perinatal infections (especially with cytomegalovirus [Barkovich and Lindan, 1994]), metabolic disorders (van der Knaap and Valk, 1991), gene mutations, and copy number variations (Stutterd and Leventer, 2014). PMG has a widely variable clinical presentation, depending on the underlying aetiology, additional syndromic features, the extent and localization of the PMG, and the presence of additional MCDs. Age at first manifestation varies from the neonatal period to late adulthood, with a large proportion of patients presenting with hemiplegia, microcephaly, global developmental delay, multiple congenital anomalies, and, most commonly, epilepsy (Guerrini et al., 1998, 1997; Parrini et al., 2009; Leventer et al., 2010; Castaño de la Mota et al., 2011; Shain et al., 2013).
Genetics
In the last decades, mutations in over 30 genes have been linked to PMG. This especially includes mutations in the tubulin genes (Stutterd and Leventer, 2014), as well as mutations in the (PI3K)-AKT pathway, in cases of PMG-associated hemimegalencephaly (Stutterd and Leventer, 2014). Bilateral perisylvian (Chang et al., 2004) PMG is mainly X-linked, though genetically heterogeneous, with some families mapping to chromosome Xq28 (Villard et al., 2002). Perisylvian asymmetric PMG, often with right hemispheric predominance, has been reported in association with chromosome 22q11.2 deletion (Robin et al., 2006). Bilateral frontal and fronto-parietal PMG has been associated with mutations of the G protein-coupled receptor gene 6 (GPR56) (Piao et al., 2005). Recent studies suggest that the pathomechanisms of brain malformation encountered in patients with GPR56 mutations may have more in common with those in congenital muscular dystrophies and cobblestone lissencephaly than in other PMG forms (Guerrini, 2010; Leventer et al., 2010). Overall, the presently identified gene mutations impact a variety of cellular mechanisms, thus preventing the ascription of PMG pathogenesis to a single pathway (Stutterd and Leventer, 2014). Despite recent genetic advances, the mechanisms by which the PMG cortex forms and the underlying aetiologies remain unknown in the vast majority of cases, rendering diagnostic and prenatal testing as well as genetic counselling particularly challenging.
Clinical spectrum
PMGs are associated with a wide spectrum of clinical manifestations ranging from merely mild language developmental delay to spastic tetraparesis, severe encephalopathy, and intractable epilepsy. More than half of patients present within the first year of life (Leventer et al., 2010). The most common features attributable to PMG are epilepsy (78%), global developmental delay (70%), and motor deficits (51%), including spastic quadriplegia in bilateral forms or hemiplegia in unilateral forms (Leventer et al., 2010). The PMG may constitute only one of several features of a metabolic disorder or of a chromosome deletion syndrome, with the clinical manifestations linked to the severity of the syndrome. Furthermore, the variety of clinical presentations reflects the extent of the PMG. Extensive bilateral or generalized PMGs are associated with an earlier age at seizure onset and increased prevalence of developmental delay prior to seizure onset (Leventer et al., 2010; Shain et al., 2013).
PMG-associated microcephaly, presenting in 50% of cases (Leventer et al., 2010), and associated malformations may be diagnosed as early as in utero, whereas PMG itself may only be diagnosed during postnatal imaging. Cardiac abnormalities in association with PMG should raise suspicion of a 22q11.2 microdeletion syndrome, whereas sensorineural hearing loss in association with PMG should raise suspicion of congenital cytomegalovirus infection. Children with PMG in the context of chromosome 22q11.2 deletion almost always present with cognitive impairment and pseudobulbar palsy, whereas the vast majority suffer from severe focal or generalized epilepsy of Lennox-Gastaut type (Guerrini, 2010).
Bilateral perisylvian PMG (Chang et al., 2004) commonly manifests with pseudobulbar palsy and isolated language delay. The full spectrum includes oromotor dysfunction and epilepsy, constituting the congenital bilateral perisylvian syndrome. This is characterized by difficulties in tongue movement, expressive speech, sucking and swallowing, excessive drooling, facial diplegia, and some degree of cognitive impairment (Stutterd and Leventer, 2014). Bilateral frontal and fronto-parietal PMG present with severe intellectual disability, motor and language impairment, and severe epilepsy, often manifesting as Lennox-Gastaut syndrome (Parrini et al., 2009). Bilateral parasagittal parieto-occipital PMG presents with mild cognitive impairment and focal onset seizures with impaired awareness (Fisher et al., 2017) with or without minor automatisms, matching an occipito-temporal origin (Guerrini et al., 1997). Unilateral hemispheric or extensive multilobar PMG, often with ipsilateral hemispheric atrophy, is predominantly sporadic. Patients present with epilepsy, hemiparesis, and mild-to-moderate developmental delay (Guerrini et al., 1998). Recent epilepsy surgery studies have reported unilateral, uni- or multilobar cases (mostly temporo-occipital or temporo-parietal), characterized by an absence of developmental delay or neuropsychological deficit, low prevalence of neurological deficit (visual field defect in only two out of eight patients), and later onset (median age: 18.5; range: 6-36) of a medically intractable focal epilepsy, amenable to epilepsy surgery (Chassoux et al., 2008; Maillard et al., 2009; Ramantani et al., 2013a).
Overall, PMG is a highly epileptogenic lesion with approximately 80% of patients eventually developing seizures, the majority within the first five years of life (Leventer et al., 2010). Nevertheless, many patients present with neurological or developmental abnormalities before the advent of epilepsy.
Epilepsy: natural course
PMG-associated epilepsy presents with variable severity, encompassing intractable cases as well as cases with favourable outcome and spontaneous remission (Guerrini et al., 1998). On the other hand, a considerable number of patients do not present with epilepsy at all. This diversity mirrors the complexity of epileptogenicity and its mechanisms in PMG.
A characteristic clinical syndrome presenting at preschool age, linked to unilateral multilobar PMG, manifests with:
- –electrical status epilepticus during sleep (ESES);
- –rare partial motor, atonic, and atypical absence seizures;
- –and cognitive deterioration in some cases.
The course may last for months to years until its cessation at puberty. In the long-term, patients either remain seizure-free or present with rare focal motor seizures during sleep (Guerrini et al., 1998). Overall, the course does not differ from that seen in ESES patients without structural brain lesions. The favourable outcome of this particular syndrome contrasts with that typically observed in PMG and other MCDs, since these rarely show spontaneous remission (Berg et al., 2009). The reasons for the emergence of ESES in PMG as opposed to other epileptogenic MCDs, such as FCD, hemimegalencephaly or SCZ, remain unclear to date. On the other hand, although ESES is quite rare, it is frequently associated with PMG. This underlines the necessity for performing sleep EEG recordings in PMG patients, particularly in the ESES age range and in the presence of focal motor seizures, attention deficit or hyperactivity (Guerrini et al., 1998). In a recent study, the occurrence of ESES was analysed in relation to the volumetric variables of the PMG hemispheres and ipsilateral thalami in 27 patients, followed prospectively over a mean of 14 years (Bartolini et al., 2016). ESES was diagnosed between five and seven years and remission occurred within two years from onset in 21%, within four years in 50%, and by age 13 years in 100% of patients. In this study, smaller thalamic and hemispheric volumes were found in PMG/CSWS with respect to PMG without CSWS, benign rolandic epilepsy and controls with headache. The volumes of the PMG hemispheres and ipsilateral thalami thus reliably led to identification of the risk of incurring ESES. The authors postulated that the PMG/ESES syndrome is linked to a cortico-thalamic malformation complex and that the assessment of hemispheric and thalamic volumes in children with PMG and epilepsy is a reliable biomarker for ESES.
In general, patients with MCDs associated with refractory epilepsy and developmental deficits (such as FCD) are candidates for resective surgery, depending on the MCD localization and extent, with several studies supporting early intervention (Maillard and Tassi, 2017; Ramantani et al., 2013b, 2013c, 2014, 2017; Kadish et al., 2018). However, favourable epilepsy prognosis in PMG-associated ESES and unclear prognosis regarding cognitive functions should discourage early surgery in this context.
In the largest PMG cohort to date, the prevalence of epilepsy did not differ significantly relative to PMG pattern, extent, localization or laterality, although patients with generalized and bilateral forms were younger at seizure onset (Leventer et al., 2010) and had a higher prevalence of developmental delay prior to seizure onset (Shain et al., 2013). The median age at presentation for the entire cohort was four months, with almost half of patients manifesting neonatal seizures. Only 7% of patients had their first seizures after the first decade and a single patient had seizure onset in the third decade. Patients with focal PMG showed a trend towards older age at seizure onset (Leventer et al., 2010; Mavili et al., 2012; Shain et al., 2013).
Magnetic resonance imaging
MRI is the imaging tool of choice in order to reliably differentiate PMG from other MCDs and to determine its extent. The “stippling” of the grey-white junction is a specific feature of PMG, not encountered in other MCDs (Leventer et al., 2010). The sylvian fissures in particular should be meticulously examined in high-quality sagittal slices, since these constitute a frequent localization of PMG. In young children, the extent of the PMG may be masked by the yet immature myelination, rendering follow-up imaging studies crucial (Barkovich, 2010). MRI features of PMG vary with the age of the patient; PMG cortex appears very thin, with multiple, very small undulations in infancy, changing to slightly thickened cortex with a slightly irregular cortex-white matter junction after myelination (Takanashi and Barkovich, 2003). The pial surface may present paradoxically as a smooth surface, due to the fusion of the molecular layer across adjacent microgyri. CT may be used additionally in order to detect calcifications associated with congenital cytomegalovirus infection, and thus clarify the aetiology of the PMG.
The heterogeneity of PMG is mirrored in its variable topographic distribution and imaging features, that relate not only to its diverse pathogenesis, but also to its distinct clinical presentation. The most common topographic pattern is perisylvian PMG, with bilateral and unilateral patterns accounting for 52% and 9% of cases in the largest PMG series to date (Leventer et al., 2010). The perisylvian cortex was the region of maximal severity in 65% patients in this series, including several patients with combined perisylvian PMG and periventricular nodular heterotopia. The percentage of perisylvian involvement in PMG, including patients with maximal involvement in other brain regions, is estimated at 80% (Hayashi et al., 2002; Leventer et al., 2010). Most other frequently observed patterns are bilateral, including generalized PMG, bilateral frontal and fronto-parietal PMG, and bilateral parasagittal parieto-occipital PMG (Barkovich, 2010; Leventer et al., 2010). In line with the hypothesis that PMG is a disorder of fissures and sulcation, perisylvian PMG affects the region around the sylvian fissure, frontal PMG is limited posteriorly by the rolandic fissure, and parasagittal parieto-occiptal PMG is centred around the parieto-occipital and calcarine sulci. Generalized PMG is observed more frequently in conjunction with peroxisomal disorders as well as with congenital cytomegalovirus infections. Rarer patterns of PMG include multifocal PMG, PMG associated with Sturge-Weber syndrome, and PMG associated with deep transmantle clefts not fulfilling the criteria for schizencephaly (Leventer et al., 2010).
Malformations such as periventricular grey matter heterotopia, ventriculomegaly, and abnormalities of the corpus callosum, brainstem, and cerebellum may be encountered in association with PMG (Wieck et al., 2005; Leventer et al., 2010). Periventricular grey matter heterotopia may occur together with periventricular or parieto-occipital PMG (Leventer et al., 2010). In parieto-occipital PMG associated with periventricular heterotopia (Wieck et al., 2005), abnormalities of the hippocampi, cerebellum or corpus callosum may occur that would be unusual for most other PMG patterns. SCZ is a specific pattern of PMG (Barkovich et al., 2005), constituting deep brain clefts that may extend through the entire cerebral mantle to communicate with the lateral ventricle. The clefts are lined with PMG, and can be thus distinguished from porencephalic cysts, which are lined by white matter or gliosis. SCZ clefts can be unilateral or bilateral and “open-lipped” (walls of the clefts do not appose) or “closed-lipped” (walls of the cleft appose and are often fused).
Ultra-high-field MRI (7T) may be a useful tool in the future, revealing more anatomical details of PMG compared with conventional 3T sequences and thus adding to its precise characterization. In a recent study, 7T MRI revealed cortical vein abnormalities, thus suggesting a role for vascular dysgenesis in PMG pathogenesis (De Ciantis et al., 2015).
Epilepsy surgery
Although 78-87% of patients with PMG are diagnosed with epilepsy (Guerrini and Filippi, 2005; Leventer et al., 2010) and 65% present a refractory course (Guerrini and Filippi, 2005), epilepsy surgery is rarely performed and results reported in MCD surgical series are rather poor (Sisodiya, 2000) (table 1). PMGs represented only 5% of MCD-related epilepsy surgery in a large monocentric study of 143 patients (Chang et al., 2011) and only 0.8% of all histopathological diagnoses in 9,523 patients submitted to epilepsy surgery in a recent multicentric study, whereas children featured much higher rates than adults (2.1% vs. 0.2%) (Blumcke et al., 2017). Most importantly, only 50% of patients reportedly achieved long-term seizure freedom (Wang et al., 2016). Epilepsy surgery in PMG raises specific issues related to:
- –the extent of the malformation that is often bilateral, multilobar or multifocal;
- –the co-existence of epileptogenic and eloquent cortex (Maillard et al., 2009; Ramantani et al., 2013a) within the PMG;
- –and the potentially favourable prognosis of some PMG-associated epilepsy syndromes (Shain et al., 2013).
The key question in epilepsy surgery for PMG-associated refractory epilepsy concerns the necessity of inclusion of the full malformation in the surgical volume. This would translate into particularly extensive resections for these bilateral, multilobar or multifocal malformations, raising the issue of overlapping with eloquent cortex. On the other hand, any surgical approach that would elude the dogma of “gross total resection for seizure freedom”, as established for FCD (Chang et al., 2011), would have to be well justified. Interestingly, several studies have reported a trend toward more right-hemispheric involvement in unilateral or bilateral asymmetric PMG patterns (Shain et al., 2013), rendering motor functions - not language - the most important consideration for resective surgery. Another central question involves the natural history of PMG-associated epilepsy, since a proportion of cases reviewed for epilepsy surgery will eventually become seizure-free with antiepileptic drugs (AEDs) alone, owing to the overall favourable prognosis of some PMG-associated clinical syndromes. However, in a recent report of 66 patients with unilateral PMG, hemiparesis and ESES (Caraballo et al., 2013), most demonstrated seizure reduction, but only three patients achieved seizure freedom with AEDs alone.
In a recent multicentre study (Shain et al., 2013), 9/61 patients underwent surgical resection following comprehensive presurgical evaluations: three had focal, five had unilateral, and one had bilateral PMG. Patients discouraged from proceeding to surgery had:
- –adequate seizure control with AEDs (n=3);
- –bilateral/multifocal seizure onsets (n=8);
- –high risk of functional deficit (n=1); or
- –extensive PMGs.
Patients who underwent epilepsy surgery had a partial or total lesionectomy, leading to seizure freedom (Engel Class I) (Engel et al., 1993) in 7/9 patients in the 12-month follow-up period. It should be noted that one of these patients featured a bilateral PMG, but with a single well-defined seizure onset. In the same study, a postsurgical deterioration of epilepsy occurred following the partial resection of an extensive PMG. A hemispherotomy was performed in 3/9 patients, although only one had a malformation extending through all four lobes. All three patients were seizure-free at follow-up. The only patient explored with intracranial recordings had a parietal PMG giving rise to epileptic spasms as well as focal seizures with left parietal onset; both seizure types ceased after a left parietal resection. Two of nine patients underwent interictal PET that showed hypometabolism, roughly correlating with the extent of the PMG in both cases. However, the hypometabolism exceeded the resection volume in the case of a focal (temporal) resection, leading to seizure freedom. Although two thirds of patients in this study had an epilepsy onset in infancy or early childhood, more than half of them underwent epilepsy surgery in adult life.
Two recent studies, based on intracranial recordings, focused on the correlations between the epileptogenic zone, the electroclinical, and neuroimaging features of PMG. In the first study (Chassoux et al., 2008), three of four patients displayed interictal high-frequency spiking within the PMG and an epileptogenic zone involving the PMG, but extending to remote cortical areas, especially to the mesial temporal structures (MTS) in two cases. In the last patient, the ictal discharge started in a region distinct from the PMG that was rapidly but secondarily involved. Three patients underwent extensive surgery including the PMG that resulted in long-term postsurgical seizure freedom. This study emphasized that:
- –despite its intrinsic epileptogenicity, the PMG was only part of a larger epileptogenic network, extending beyond the MRI-detectable malformation;
- –and postsurgical seizure freedom could be obtained, provided that a large resection including the PMG was feasible.
In the second study (Ramantani et al., 2013a), the epileptogenic zone was limited to the MTS in one patient, fully sparing the PMG. It primarily involved the MTS and secondarily involved part of the PMG in two patients, and primarily involved both the MTS and the entire PMG in the last patient. This study demonstrated that in unilateral regional multilobar (mainly infrasylvian) PMG:
- –the intrinsic epileptogenicity of the PMG was heterogeneous;
- –the epileptogenic zone involved remote but functionally connected cortical areas, namely the MTS;
- –the PMG was occasionally and partially involved in the epileptogenic zone;
- –a surgical resection based on intracranial recordings, sparing part or the totality of the PMG, led to long-term postsurgical seizure freedom (Ramantani et al., 2013a).
In a larger case series of surgery for PMG-associated refractory epilepsy (Wang et al., 2016), six of 12 patients achieved long-term seizure freedom and another four significant seizure reduction after focal resections in eight and hemispherectomy in four cases. In accordance with previous studies, this cohort featured a childhood onset of epilepsy in 83% of patients, whereas one patient presented with ESES and another patient had infantile spasms. Eight of 12 patients had unilateral PMG and four bilateral (two with bilateral perisylvian and two with bilateral occipital involvement), with an involvement of the perisylvian region in half of the cases. In additional to PMG, eight of 12 patients featured further MCDs, including three patients with hemimegalencephaly, two patients with FCD, two patients with heterotopia, and one patient with closed-lip SCZ. Scalp EEG findings were not useful in most cases, except for providing seizure onset lateralization in two of four patients with bilateral PMG. Three of eight patients with unilateral PMG had poorly lateralized seizures on scalp EEG and one presented with ESES. With the exception of the three patients who underwent hemispherectomy, all other patients had either long-term intracranial recordings or intraoperative electrocorticography (ECoG) for further delineation of their seizure foci. These invasive recordings were deemed useful for guiding resective surgery in patients with diffuse and non-lateralizing scalp EEG findings, including a patient with normal scalp EEG. Interestingly, intracranial recordings demonstrated good concordance with all four lateralizing scalp EEG recordings. With intracranial EEG, seizure onset was focal in seven and diffuse in two patients; none of the patients with diffuse onset achieved postsurgical seizure freedom. The limitations of this study arise from the lumping together of extra- and intraoperative intracranial EEG recordings, considering the limitations of the intraoperative setting as to the spontaneous recording of seizures. As expected in a cohort including five hemispherectomies as the final surgery, postoperative neurological deficits included contralateral hemiparesis in six and hemianopia in four patients, and language and cognitive delay in another four patients. The study reported a trend for more favourable postsurgical seizure outcomes following the full resection of the PMG, but not of other associated MCDs. This observation is to be treated with caution, due to:
- –the limited number of patients (n=12);
- –and the lumping together of focal and hemispheric surgery, especially since the study included three patients with PMG-associated hemimegalencephaly.
These three epilepsy surgery studies, drawing partly or entirely from invasive recordings, have drawn attention to the potential of intracranial and/or intracerebral recordings to delineate tailored resections with favourable postsurgical seizure outcomes, in spite of the extensive MRI lesions and EEG abnormalities in patients with PMG-associated epilepsy.
A more recent study including 64 patients with PMG-associated refractory epilepsy undergoing presurgical evaluation (Cossu et al., 2016), that led to surgical treatment in 24 patients, showed a higher rate of seizure freedom in the operated group compared to the medical treatment group. This finding suggests that, in a subset of selected patients with PMG-related epilepsy, surgery may represent an efficient therapeutic option. Indeed, at last follow-up visit, 67% (18 of 24) of surgical patients had remained seizure-free; six after withdrawal of antiepileptic drugs. These favourable results were achieved after SEEG evaluation in 58% of surgical patients, demonstrating the strong contribution of SEEG to the identification of lesional as well as extralesional areas involved in the epileptogenic zone and to the delineation of potentially overlapping eloquent cortex. The authors underlined the advantages of SEEG compared to (1) intracranial recordings with subdural electrodes that preclude sampling of PMG cortex in areas such as the deep banks of the schizencephalic clefts, as well as (2) intraoperative electrocorticography that has clear limitations for functional mapping. Finally, this cohort included five patients who underwent hemispherectomy with excellent seizure outcomes, thus underlining that hemispheric PMG patients can be excellent candidates for hemispherectomy, particularly in the presence of contralateral hemiparesis. It should, however, be noted that, in this study, patients selected for surgical treatment had almost exclusively unilateral MRI lesions, in contrast to those excluded from surgery (Cossu et al., 2016).
The so far largest multi-centric cohort of PMG-related drug-resistant epilepsy patients undergoing SEEG and/or epilepsy surgery to date (Maillard and Tassi, 2017) clearly displayed a variable degree of PMG involvement in the epileptogenic zone, often combined with the additional or predominant involvement of remote cortical areas, as identified in 49 SEEG investigations. In this study, the epileptogenic zone involved only part of the PMG in most cases (60%), with additional remote cortical areas in 18%. The other constellations comprised, in decreasing order of frequency, an epileptogenic zone involving:
- –the entire PMG without additional remote cortical areas;
- –the entire PMG and remote cortical areas;
- –and only remote cortical areas.
The partial or lack of involvement of the PMG in the epileptogenic zone in 70% cases in this study (Maillard and Tassi, 2017) stands in stark contrast to the more straightforward concordance of the malformation to the epileptogenic zone encountered in FCD type II, the most common surgically remediable malformation (Guerrini et al., 2015). Furthermore, this study highlighted the involvement of remote cortical areas in 43% cases, 12 of 21 relating to the mesial temporal structures, due to either an associated -and potentially epileptogenic- malformation (hippocampal malformation, parahippocampal nodular heterotopia, and schizencephaly) or to secondary epileptogenesis. These observations clearly underline the need for a different strategy in PMG-related drug-resistant epilepsy compared to other MCDs, namely one that is not predominantly MRI- but rather SEEG-oriented (Maillard and Tassi, 2017). In addition to SEEG-guided surgery, this multicentric study provided data on nine patients undergoing direct epilepsy surgery: four of these nine patients underwent hemispherectomy for hemispheric PMG. Young children with a hemispheric PMG, multifocal unilateral seizure onset, and presurgical hemiparesis who eventually undergo hemispherectomy differ to a great extent from patients undergoing SEEG-guided focal or regional corticectomy. At the other end of the spectrum, in very selected cases of small-sized unilobar PMG with fully concordant electroclinical findings, direct resection of the entire PMG may also be a reasonable option, provided that there is no electroclinical or PET evidence of remote cortical involvement.
Presurgical evaluation for polymicrogyria
Illustrative case
We report the illustrative case of a 27-year-old, right-handed woman who presented with a first seizure at the age of 18. Initial ictal symptoms comprised a dreamy state and a warm ascending sensation. She was able to warn that she was going to have a seizure, and manifested with an early flush and loss of consciousness. Later, in the course of the seizure, she had right upper limb automatisms and left upper limb tonic posturing. Mean seizure duration was 60 seconds. On average, the patient suffered 6-8 focal seizures per month and two secondary generalized tonic-clonic seizures per year.
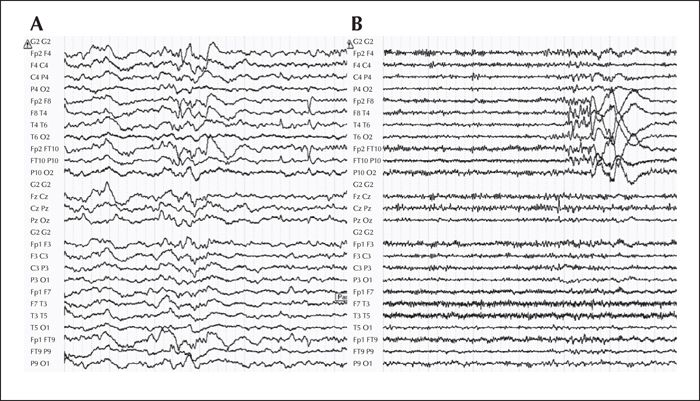
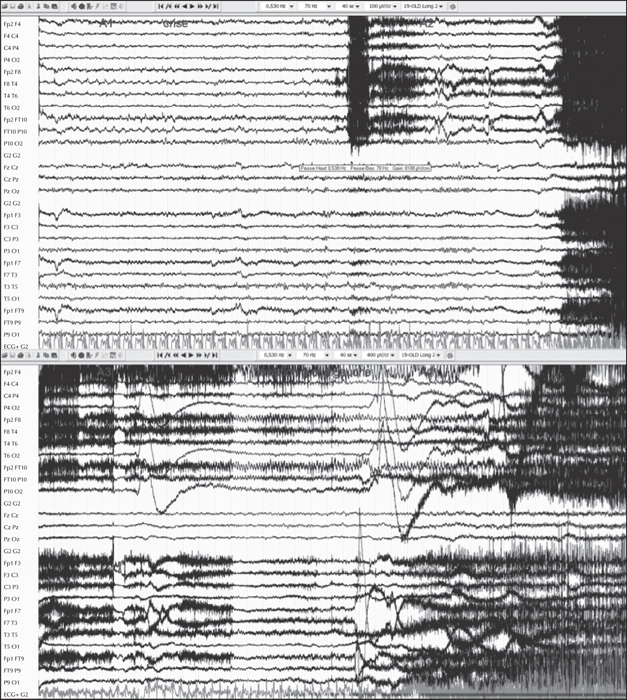
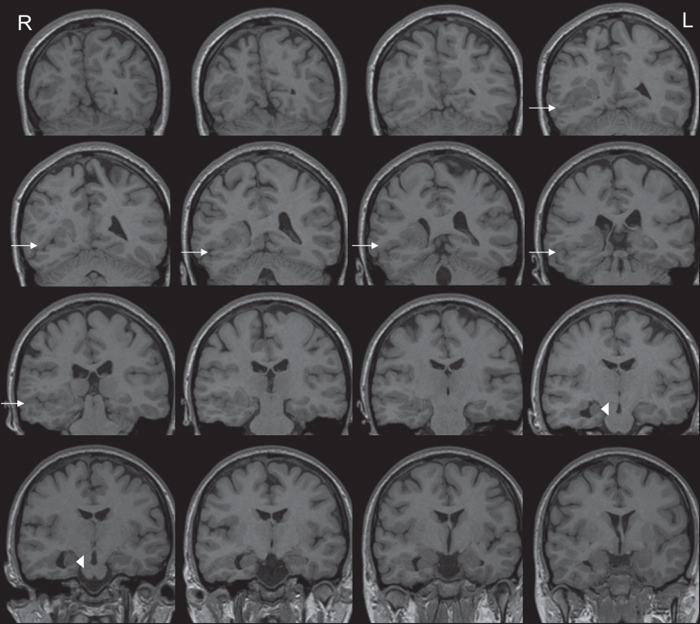
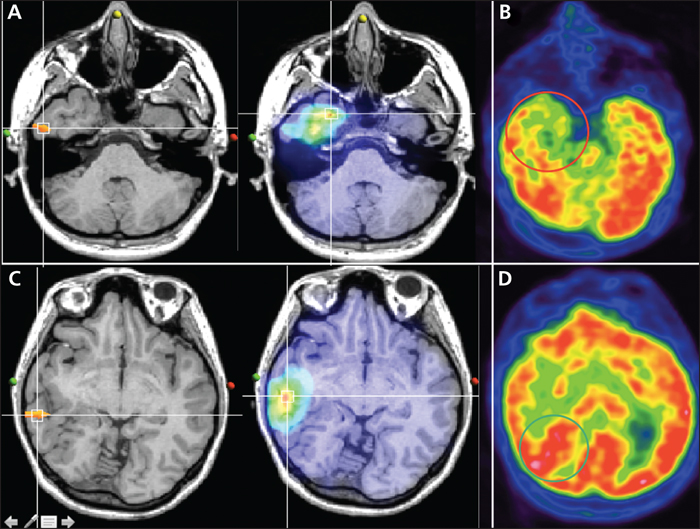
Interictal EEG showed right temporal theta activity and two types of focal paroxysms: sharp waves, maximum at electrodes FT10 and F8 (figure 1A) and 8-Hz polyspikes, followed by a high-amplitude delta wave, projecting at electrodes T4, T6 and FT10 (figure 1B). The first ictal EEG changes were characterized by a right hemispheric flattening, followed by a focal right temporal (FT10, F8, T4, FP10) alpha (8-Hz) rhythmic discharge, and ending with an irregular high-amplitude delta activity (figure 2). Cranial MRI showed widespread right occipito-parieto-temporal PMG surrounding a closed lip schizencephaly (SCZ) that extended from the lateral occipito-temporo-parietal junction to the right lateral ventricle. Associated malformations comprised a periventricular nodular heterotopia and a right hippocampal malrotation (figure 3). Electrical source imaging (ESI) identified two distinct sources: medial and lateral temporo-polar, remote from the PMG (figure 4A), and lateral temporal in the middle part of the PMG (figure 4C). FDG-PET showed significant hypometabolism in the right temporal pole, consistent with the first source (figure 4B), whereas the most posterior part of the PMG lining the schizencephalic cleft appeared isometabolic (figure 4D).
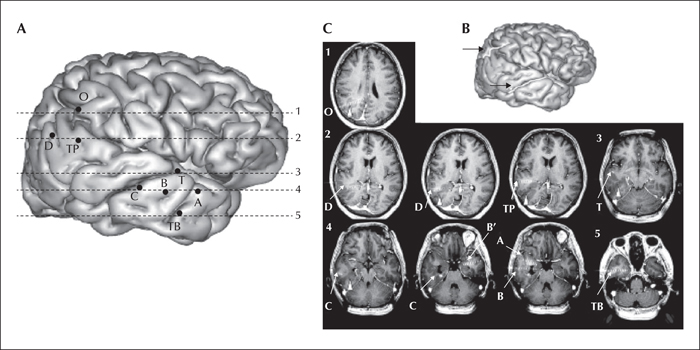
The goal of SEEG was to delineate the epileptogenic zone. The electrode implantation sites were chosen according to non-invasive data collected during the earlier phase of the investigation. Eight electrodes were placed in the right hemisphere targeting the amygdala, hippocampus, parahippocampal gyrus, the PMG cortex along the middle and superior temporal gyrus, the PMG cortex lining the anterior and posterior lip of the SCZ, and the nodular heterotopias (figure 5). Two types of paroxysmal events (figure 6) were identified on interictal recordings:
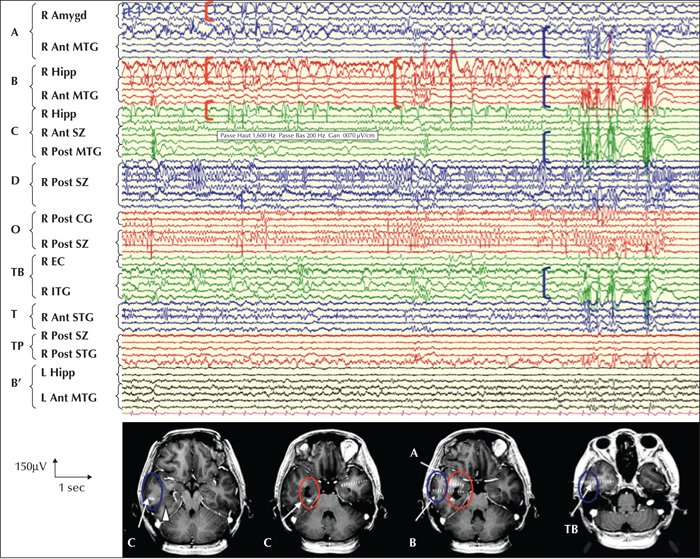
- –abundant spike-wave paroxysms outside of the PMG, simultaneously recorded in the amygdala (A; internal leads) and hippocampus (B and C, internal leads), sometimes propagating to the anterior middle temporal gyrus (A and B, external leads);
- –and bursts of polyspike-wave paroxysms recorded in the anterior part of the PMG involving the middle and inferior temporal gyri (B, C and TB, external contacts), propagating to the anterior superior gyrus (T) and rarely -in the immediate postictal phase- to the temporo-parieto-occipital junction in the posterior part of the SCZ (D, external contacts).
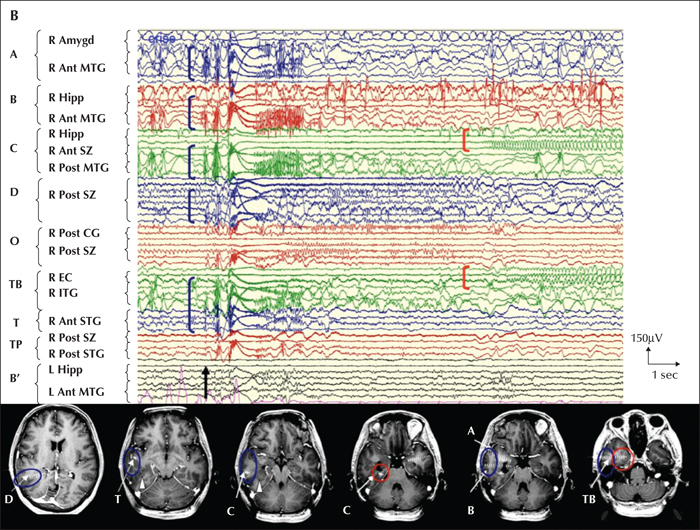
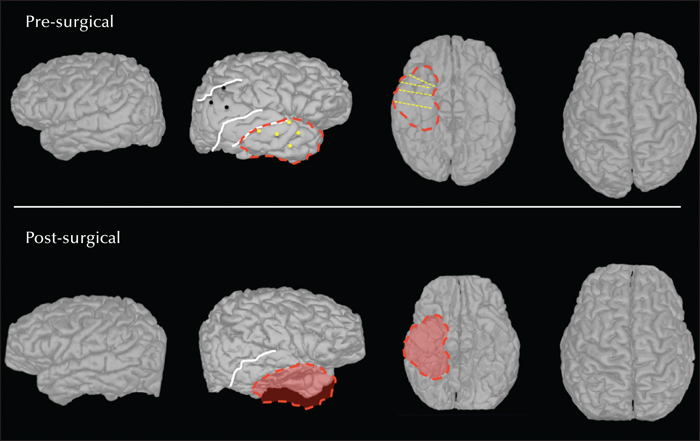
Two types of spontaneous seizures were recorded: the first type (most frequent, corresponding to the usual clinical seizures) initially involved the MTS, propagating secondarily to the temporal PMG cortex (anterior and posterior mesial temporal gyrus, anterior superior temporal gyrus), but sparing the most posterior part of the PMG cortex (figure 7A). The second seizure type initially involved part of the PMG cortex and secondarily spread outside of the PMG to the MTS (figure 7B). Electrical stimulation within the PMG-associated part of the epileptogenic zone did not elicit seizures, whereas electrical stimulation in the remote cortical area (MTS) involved in the epileptogenic zone did elicit habitual seizures. The resection included the MTS and the anterior part of the PMG (figure 8). The patient has remained seizure-free so far (eight years post-surgery), with a partial reduction in her drug treatment from a combination therapy to low-dose monotherapy.
Indications for presurgical evaluation
Although most patients with PMG present with epilepsy in the first years of life, presurgical evaluation and, eventually, surgical intervention, reportedly occur with a latency of over 10 years (Wang et al., 2016). This may be partly attributed to the prospect of seizure freedom with AEDs alone that, unfortunately, holds true only for a minority of patients (Caraballo et al., 2013) as well as to the inherent difficulties in dealing with these often very extensive malformations. On the other hand, surgery has been shown to provide seizure freedom for carefully selected candidates (Chassoux et al., 2008; Maillard et al., 2009; Ramantani et al., 2013a; Shain et al., 2013; Wang et al., 2016) and timely intervention, particularly in children, may help to prevent the damage caused by seizures and AEDs to the developing brain and thus offer developmental and cognitive benefits (Işik et al., 2007). Taken together, the three studies assessing the relationship between the epileptogenic zone defined by invasive EEG recordings, the electroclinical features, and the neuroimaging features of the PMG showed that nine of nine patients with unilobar or unilateral multilobar PMG, who underwent epilepsy surgery, were seizure-free or had rare residual seizures (Engel Class I or II), whereas only five of 10 (50%) with perisylvian or multifocal PMG experienced such a significant improvement. This difference is statistically significant (p=0.032). In spite of the obvious limitation arising from the small number of cases (n=14), these observations support the view that epilepsy surgery should be considered as a valid therapeutic option in cases of unilobar or unilateral bilobar PMG with intractable epilepsy. They also suggest that patients with perisylvian or bilateral PMG should not be excluded from presurgical evaluation with invasive recordings and subsequent epilepsy surgery, provided that electroclinical features are consistent with a single epileptic focus.
Non-invasive evaluation
PMG patients form a group that is very difficult to approach surgically, usually requiring multimodal investigations for accurate localization of the epileptogenic zone.
Genetic testing
Although genetic testing is not strictly part of the presurgical investigation in itself, it is important to assess the presence of associated genetic factors before considering presurgical evaluation. It remains unclear whether the specific genetic aetiology underlying at least some PMG cases can influence postsurgical seizure outcomes and to what extent. Furthermore, it is as yet unresolved whether certain genotypes are associated with a positive response to AEDs and a favourable long-term seizure outcome and should thus be excluded from presurgical evaluation and surgical treatment. Mutations in the (PI3K)-AKT pathway should be investigated in cases of PMG-associated hemimegalencephaly (Stutterd and Leventer, 2014), as well as 22q11.2 deletions in perisylvian PMG and GPR56 mutations (Piao et al., 2005) in bilateral frontal and fronto-parietal PMG.
Interictal & ictal EEG
PMG has been identified as a symptomatic cause of ESES (Guerrini et al., 1998), thus emphasizing a particular pattern of sleep activation distinct from other malformations, such as FCD type 2. Therefore, both awake and sleep recordings should be performed in PMG patients. Overall, scalp EEG findings in PMG-associated epilepsy have been reported to be diffuse and generalized, thus questioning the localizing value of EEG for presurgical evaluation, other than pointing to the correct lateralization of seizure onset in bilateral malformations (Shain et al., 2013; Wang et al., 2016). This is in contrast to simultaneous EEG/MEG studies (Bast et al., 2005) that have reinforced the correlation between lesion topography and EEG correlates. This discrepancy may be attributed to the number of sensors in different studies, linked to a variable resolution of EEG patterns.
In a retrospective study including 40 PMG patients, 55% presented with no EEG abnormalities at all (Teixeira et al., 2007). In this study, the severity of EEG -and clinical- features correlated with the extent of cortical involvement. In perisylvian PMG, epileptic discharges predominated in fronto-temporal regions, while other EEG abnormalities manifested in centro-temporal regions. Ten percent of patients presented with focal electrical status in wakefulness, evolving into ESES in sleep. Altogether, 20% of patients had activation of epileptic discharges in sleep. In a follow-up study considering six PMG patients (Teixeira et al., 2009), the authors further characterized the constellation of a focal electrical status, presenting with continuous epileptic discharges over a focal area in wakefulness, and becoming bilateral and synchronous during sleep. Interestingly, continuous and rhythmic epileptiform discharges on intraoperative ECoG (Palmini et al., 1995), as well as on scalp EEG (Gambardella et al., 1996), have been previously acknowledged as the EEG signature of cortical dysplastic lesions. The authors (Teixeira et al., 2009) proposed that the PMG-associated focal electrical status may be distinguished by the high degree of activation in sleep with the emergence of bilateral and synchronous discharges, whereas the FCD-specific rhythmic epileptiform discharges show no clear modification of their electrical field during sleep.
In the three anatomo-electro-clinical concordance studies with detailed results of invasive recordings (Chassoux et al., 2008; Ramantani et al., 2013a; Wang et al., 2016), eight of 10 patients with unilobar or unilateral bilobar PMG had focal or regional scalp EEG ictal discharges while four of seven patients with extended perisylvian or multifocal PMG presented with diffuse or hemispheric ictal discharges. This illustrates that, in most cases, the visual analysis of ictal scalp EEG is concordant on a regional level with the extent of the PMG.
FDG-PET
In two of three recent studies mentioned above (Chassoux et al., 2008; Ramantani et al., 2013a), FDG-PET results were reported in seven cases in relation to the PMG and the epileptogenic zone, as defined by SEEG. Focal hypometabolism included the PMG in three out of seven patients. In the remaining four cases, the PMG was isometabolic. In all cases, hypometabolism included cortical areas outside of the PMG, namely the MTS, thus matching the epileptogenic zone, as defined by SEEG. However, in the so far largest multi-centric study of PMG-related drug-resistant epilepsy patients undergoing SEEG and/or epilepsy surgery (Maillard and Tassi, 2017), the full concordance of focal FDG-PET hypometabolism with the epileptogenic zone amounted to a mere 32%. The lower concordance rate of FDG-PET findings to the epileptogenic zone observed in this study might be related to the inclusion of cases with variable PMG localizations, other than the predominantly infrasylvian cases addressed in the two previous studies (Chassoux et al., 2008; Ramantani et al., 2013a). Interestingly, in this large multi-centric study, six of 12 cases with an epileptogenic zone involving remote cortical areas related to the MTS were highlighted by FDG-PET. Overall, these findings suggest, in line with previous observations (Burneo et al., 2004a), that FDG-PET can be useful for devising the SEEG implantation scheme in PMG patients, particularly for those with infrasylvian PMG and electroclinical evidence of MTS involvement.
SPECT
In the only SPECT study on PMG to date (Wichert-Ana et al., 2008), 17 patients with PMG-associated epilepsy underwent activated (ictal) and baseline (interictal) SPECT as part of their presurgical evaluation. These were further submitted to voxel-based analysis and subtraction and co-registered to MRI (SISCOM: Subtraction Ictal SPECT Co-Registered to MRI). SISCOM images showed pronounced and statistically significant hyperperfusion overlapping with the PMG lesion in all but one patient. This patient already demonstrated moderate hyperperfusion of the PMG lesion on the interictal SPECT that resulted in a lower perfusion difference compared to the ictal SPECT. The topography and extent of the observed hyperperfusion varied according to the topography and extent of the PMG. Localized hyperperfusion was observed in three of four patients with posterior cortex PMG, four out of six patients with frontal PMG, and in only one out of six patients with perisylvian PMG. In three of six patients with perisylvian PMG, hyperperfusion was lateralized. Overall, perisylvian PMG exhibited either non-lateralizing/-localizing or, in some cases, merely lateralizing hyperperfusion, while frontal and posterior PMG exhibited more localized findings. Interestingly, the only patient in this study to feature bilateral diffuse PMG had lateralizing SISCOM findings. Based on the postsurgical outcomes of the nine patients with resective surgery (focal resections or hemispherotomy), this study supports the utility of SPECT as a valuable tool in epilepsy surgery for PMG-associated epilepsy, predicting seizure freedom in the individual patient. SISCOM findings were partially or completely within resection margins or the disconnected hemisphere in four patients; three of four patients obtained postsurgical seizure freedom. SISCOM findings were outside the resection margins in all five patients with recurrent seizures. Based on these findings, the authors discussed a possible role of PMG cortex either as part of the epileptogenic zone or as ictal spread and the symptomatogenic zone. The utility of SPECT in presurgical evaluation of PMG-associated epilepsy is also underlined in a single case (Shain et al., 2013) of bilateral perisylvian PMG, in which SPECT pointed to the right hemisphere in a child who eventually underwent hemispherotomy with excellent results.
Electromagnetic source imaging (high-resolution EEG and MEG)
ESI is a very sensitive and reliable tool that contributes to the localization of the epileptogenic zone in MCDs (Rikir et al., 2014), especially in PMG (Maillard et al., 2009; Ramantani et al., 2013a). High-resolution (HR-) EEG has proven superior to MEG, due to the predominant radial orientation of generators, resulting from the particular anatomy of the multiple excessive small gyri on the convexity (Guerrini and Filippi, 2005). It is particularly useful in selecting the appropriate targets for SEEG exploration, especially in cases of extensive PMG with its heterogeneous epileptogenicity and functionality.
Magnetic source imaging (MSI) of evoked potentials has also been applied to PMG as well as to other MCDs, in order to shed some light on the potential for functional reorganization of the primary sensory cortex (Burneo et al., 2004b). In this study, it was demonstrated that somatosensory function remains localized in the polymicrogyric rolandic cortex, as long as anatomy is not distorted by a schizencephalic cleft.
EEG-fMRI and fMRI
EEG-fMRI is a useful tool for the non-invasive assessment of interictal spike generators. The heterogeneous and multifocal epileptogenicity of the malformation was demonstrated in nine patients with perisylvian (n=6), bi-frontal (n=1), and parieto-occipital (n=2) PMG (Kobayashi et al., 2005). For both activations and deactivations, only part of the malformation was involved concomitantly with cortical areas outside of the PMG.
In accordance with this heterogeneous epileptogenicity, several fMRI studies have demonstrated that PMG may still harbour function, including primary somatosensory and motor functions as well as language in cases of perisylvian PMG extending to frontal and rolandic cortex (Araujo et al., 2006; Vitali et al., 2008). A functional reorganization extending beyond the limit of the PMG may be encountered (Vitali et al., 2008), although it has not been systematically observed (Nikolova et al., 2015). Combined fMRI and transcranial magnetic stimulation studies in patients with PMG and hemiparesis have demonstrated that PMG cortex can show various degrees of participation in motor functions; ipsilateral corticospinal projections from the contralesional hemisphere to the paretic hand, corticospinal projections to the paretic hand originating in the PMG cortex, and bilateral motor representation were all recorded (Staudt et al., 2004). This heterogeneity in the reorganization patterns of cortical function in the context of PMG has been ascribed to differences in the severity of disruption.
Normal cortical responses and organization of visual areas have been observed in patients with bilateral parieto-occipital PMG, using phase-encoded retinotopic mapping analysis (Dumoulin et al., 2007). Interestingly, in all but one case, the reconstructed primary visual cortex fully overlapped with the PMG, supporting the notion that PMG cortex is not only organized in a normal fashion, but is also actively involved in processing of visual information and contributes to normal visual perception. However, caution should be taken with the generalization of any findings regarding cortical organization in PMG patients, due to the causal heterogeneity and hence the high degree of histopathological variability (Guerrini, 2010).
More recently, an fMRI connectomics study showed an abnormal network topology that could account for the intrinsic and remote excitability (Sethi et al., 2016), but the clinical relevance of these novel findings remains to be evaluated.
Overall, fMRI is mandatory in the presurgical evaluation to assess the functionality of PMG cortex and the potential functional re-organization of motor, visual, and language systems. The functions investigated by fMRI should match the localization of the PMG (sensorimotor, visual, and language).
How to proceed to the delineation of the resection
SEEG exploration strategies
The SEEG implantation scheme primarily relies on the anatomo-electro-clinical hypothesis. This is particularly relevant in PMG, since the epileptogenicity of the MR-visible malformation is heterogeneous, whereas remote cortical areas or associated malformations may play a decisive role in seizure initiation. Therefore, SEEG schemes generally sample large parts of the PMG and of remote cortical areas that are functionally connected to the malformation, such as the MTS in unilateral infra-sylvian PMG. The choice of the regions of interest is also guided by FDG-PET and ESI/MSI findings that help to focus the electroclinical hypothesis derived from scalp video-EEG recordings (Sethi et al., 2016). The SEEG scheme is also devised for the purpose of functional mapping and should especially target parts of the PMG in the vicinity of the suspected seizure onset zone, as well as those that were shown to harbour functions on fMRI.
SEEG recordings
Interictal findings
Interictal epileptic activity may be completely absent from part or even the entirety of the PMG, as reported in two recent studies; in one of four cases (Chassoux et al., 2008) and two of four cases (Ramantani et al., 2013a). Intralesional epileptic activity typically consists of high-frequency (>15-c/s), high-amplitude polyspikes (figure 6). Less specific patterns, such as sharp waves, can also be observed. Spikes and sharp waves may also be observed outside of the PMG in remote cortical areas, such as the MTS. These intra and extra-lesional networks may be either independent or synchronized.
Ictal findings
When the seizure onset is located within the PMG, the typical pattern is a pre-ictal polyspike discharge, followed by a fast low-voltage activity (figure 7B). When the seizure onset is located outside of the PMG, e.g. in the MTS (Ramantani et al., 2013a), the typical pattern is a fast low-voltage discharge. The seizure onset zone spared part or the totality of the PMG in 10 of 19 operated patients with eight of 10 patients achieving significant improvement (Engel Class I or II) (Chassoux et al., 2008; Ramantani et al., 2013a; Shain et al., 2013; Wang et al., 2016).
Intracortical electrical stimulation
In contrast to FCD, intralesional electrical stimulation rarely reproduces spontaneous seizures (reported in one of eight patients in recent studies [Chassoux et al., 2008; Ramantani et al., 2013a]). Stimulation in the remote cortical areas involved in the epileptogenic zone, especially in the MTS, elicited usual seizures in four out of eight patients. Therefore, electrical stimulation (using low [1-Hz] and high [50-Hz] frequency) should encompass both the PMG and suspect remote cortical areas. The absence of seizures elicited by electrical stimulation of the PMG does not rule out its involvement in the epileptogenic zone, since this primarily relies on the recording of spontaneous seizures.
Post-operative outcome
The available data on seizure outcomes following epilepsy surgery for refractory PMG-associated epilepsy are overall sparse and, to some extent, contradictory, most likely due to the inclusion of diverse epilepsy syndromes, investigative procedures, and surgical resections (Chassoux et al., 2008; Wichert-Ana et al., 2008; Maillard et al., 2009; Ramantani et al., 2013a; Shain et al., 2013; Cossu et al., 2016; Wang et al., 2016). The several reported case series so far have each included four to 12 patients, investigated using variable methodology, including PET (Chassoux et al., 2008; Ramantani et al., 2013a; Shain et al., 2013), SPECT (Wichert-Ana et al., 2008), or neither (Wang et al., 2016), derived solely from long-term invasive EEG recordings for the delineation of the epileptogenic zone (Chassoux et al., 2008; Ramantani et al., 2013a) or not (Wichert-Ana et al., 2008; Shain et al., 2013; Wang et al., 2016), with a variable proportion of hemispherectomies and focal resections. Accordingly, reported postsurgical seizure freedom figures range from 25% (3/12) to 78% (7/9) in these case series and no specific predictors of seizure freedom could be identified. Nevertheless, the most recent of these case series (Cossu et al., 2016) showed a higher rate of seizure freedom in the operated group compared to a medical treatment group, suggesting that, in a subset of selected patients with PMG-related epilepsy, surgery may represent an efficient therapeutic option.
In the first large multi-centric cohort of 58 PMG-related drug-resistant epilepsy patients undergoing SEEG and/or epilepsy surgery (Maillard and Tassi, 2017), seizure freedom after ≥two years of follow-up was achieved in 72% of patients, establishing the overall favourable outcomes of surgical treatment in selected candidates within this challenging patient group. The high rates of seizure freedom in this large cohort, with 77% patients undergoing SEEG-guided epilepsy surgery, reflect the potential of SEEG to delineate tailored resections in PMG-related epilepsy with favourable postsurgical seizure outcomes, in spite of the extensive MRI lesions and EEG abnormalities. Indeed, the unfavourable 33-50% rates of seizure freedom following PMG-related epilepsy surgery stem from patient series with 25% and 33% invasive recordings (Wang et al., 2016; Wichert-Ana et al., 2008), whereas a recent study relying on SEEG to delineate the EZ in 58% patients reported a more favourable 67% rate of seizure freedom (Cossu et al., 2016). It should be noted that, in the multi-centric study (Maillard and Tassi, 2017), the full resection of PMG cortex was not associated with superior seizure outcomes compared to the partial or no resection of PMG cortex. This supports the notion that a surgical resection based on intracranial recordings, sparing part of or the entire PMG, can lead to long-term seizure freedom. Seizure outcomes in this study (Maillard and Tassi, 2017) remained unaffected by the extent of the malformation, ranging from 75% to 80% for unilobar vs. multilobar vs. perisylvian vs. hemispheric PMG. This observation underlines that, in selected cases, the presence of an extensive PMG does not necessarily call for an extensive resection with possible functional losses. However, this study (Maillard and Tassi, 2017) also pointed out that SEEG should be indicated with caution in bilateral PMG, considering the reduced chance of proceeding to successful epilepsy surgery that clearly concerns only cases with a unilateral epileptogenic zone. Finally, this large multi-centric study was the first to highlight the prognostic implication of epilepsy duration, specifically in PMG-related drug-resistant epilepsy, on long-term seizure outcomes (Maillard and Tassi, 2017). The negative effect of epilepsy duration may be related to a progressive establishment of epileptogenic networks within the PMG and the associated malformations over time, culminating in secondary epileptogenesis.
Conclusion
PMG-associated refractory epilepsy warrants a detailed presurgical evaluation, including non-invasive investigations such as scalp EEG, volumetric MRI and PET or SPECT scans, fMRI, and when available HR-EEG- or MEG-derived source analysis, and eventually, invasive investigations, preferably with SEEG, in order to accurately delineate the epileptogenic zone, since the EZ may only partly overlap with the PMG or include only remote cortical areas. Favourable results regarding seizure freedom are feasible in a large proportion of patients and the extent of visible malformation should not deter from exploring the possibility of epilepsy surgery. Recent data support the early consideration of epilepsy surgery in patients with PMG-related drug-resistant epilepsy. Still, caution is needed in setting the indication for surgery in children, since some forms of PMG-associated epilepsy may entail a favourable prognosis with spontaneous recovery at puberty.
Supplementary data.
Summary didactic slides are available on the www.epilepticdisorders.com website.
Disclosures
None of the authors have any conflict of interest to declare.