European Journal of Dermatology
MENUThe Dutch Hyacorp® filler catastrophe: new EU legislation will prevent this from happening again Volume 32, issue 5, September-October 2022
- Key words: adverse events, complications, cosmetic, filler, hyaluronic acid, Hyacorp
- DOI : 10.1684/ejd.2022.4328
- Page(s) : 584-8
- Published in: 2022
Background
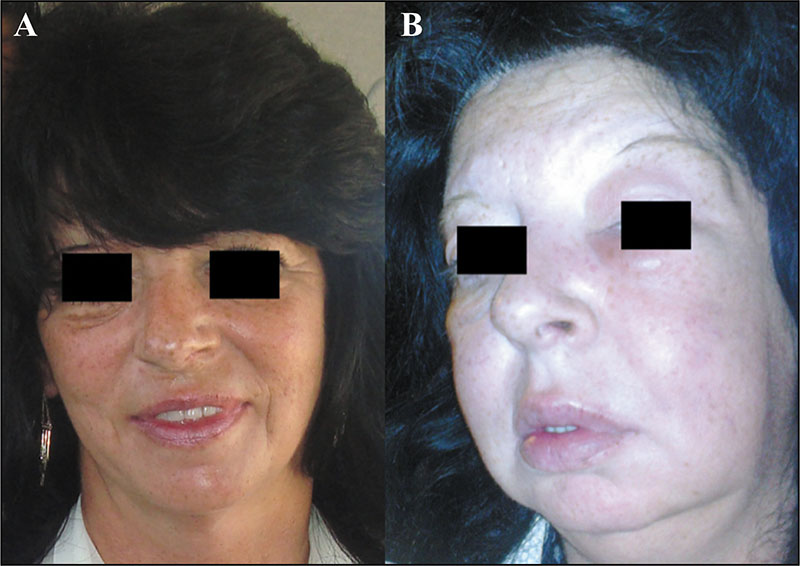
In 2014, the hyaluronic acid-based fillers, Hyacorp-1000 and Hyacorp H-S (H-800), were withdrawn from the Dutch market after concerns about their safety
Objectives
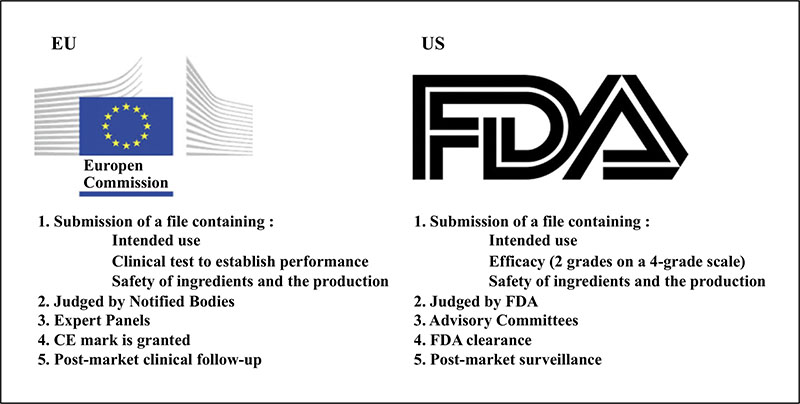
To determine the most plausible factors for the increased number of adverse events, either patient-related factors or those inherent to the filler itself. We also assessed how new European legislation will affect the approval process for new fillers and improve related safety issues
Materials & Methods
A total of 42 patients–37 women (88%) and five men (11%)–were included. Patients were separated into three groups: 13 patients injected with Hyacorp-1000 and Hyacorp H-S (H-800) who had reported inflammatory adverse events; 12 injected with Hyacorp-1000 and Hyacorp H-S (H-800) who had not reported inflammatory adverse events; and 17 injected with other HA fillers who had reported inflammatory adverse events
Results
Patients treated with Hyacorp-1000 and Hyacorp-S (H-800) who reported adverse events were significantly older than those in the Hyacorp-1000 and Hyacorp-S (H-800) group without adverse events, and the filler remained in situ for significantly longer than in patients who had adverse events related to another HA filler
Conclusion
Hyacorp-1000 and Hyacorp-S (H-800) filler is associated with an increased chance of developing adverse events compared to other HA fillers, probably because it remains in the body for a longer period of time. The upcoming legislative EU update of the Medical Device Regulation (MDR) will prevent unsafe filler from entering the EU market and will enable issues related to safety to be identified much earlier