European Journal of Dermatology
MENUStaging of primary cutaneous follicle centre B-cell lymphoma: bone marrow biopsy, CD10, BCL2 and t(14;18) are not relevant prognostic factors Ahead of print
- Key words: primary cutaneous follicle centre B-cell lymphoma, PCFCCL, bone marrow biopsy, BCL2, CD10, t(14, 18), staging
- DOI : 10.1684/ejd.2018.3489
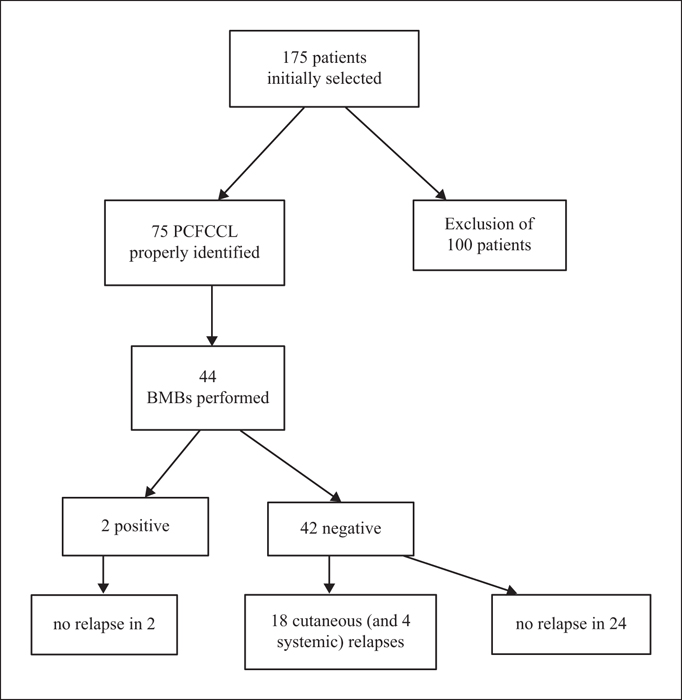
Background: There is a certain degree of controversy as to whether bone marrow biopsy is required during the staging procedures for primary cutaneous follicle centre B-cell lymphoma (PCFCCL). Objectives: Firstly, to determine extra-cutaneous involvement at initial diagnosis, in particular, based on bone marrow biopsy, and secondly, evaluate the phenotypic features associated with extra-cutaneous involvement during follow-up (in particular, the predictive value of BCL2 and CD10 coexpression and identification of t[14;18] in skin lesions, as well as bone marrow biopsy involvement at initial staging) in a cohort of patients with PCFCCL. Materials & Methods: A bicentric retrospective study was established to investigate 75 cases of PCFCCL, for which 44 bone marrow biopsies were performed. Results: Two of 44 (5%) patients had bone marrow involvement. These two patients had no relapse during follow-up, either cutaneous or extra-cutaneous. BCL2 staining in B cells was positive in 39/75 (52%) cases and CD10 was positive in 39/73 (53%). Only 4/26 (15%) cases showed t(14;18) based on fluorescence in situ hybridisation. Conclusions: Our study combined with data from the literature suggests that systematic bone marrow biopsy at initial staging for putative PCFCCL is not to be recommended. Moreover, BCL2 or CD10 expression does not currently represent a reliable basis to introduce significant changes in initial therapy or the follow-up strategy.