European Journal of Dermatology
MENUMaintenance effect of a once-weekly regimen of a Selenium Disulfide-based shampoo in moderate-to-severe scalp seborrheic dermatitis after initial treatment with topical corticosteroid/salicylic acid Volume 33, supplement 1, Mars 2023
- Key words: selenium disulfide, scalp seborrheic dermatitis, scalp, corticosteroids
- DOI : 10.1684/ejd.2023.4401
- Page(s) : 13-8
- Published in: 2023
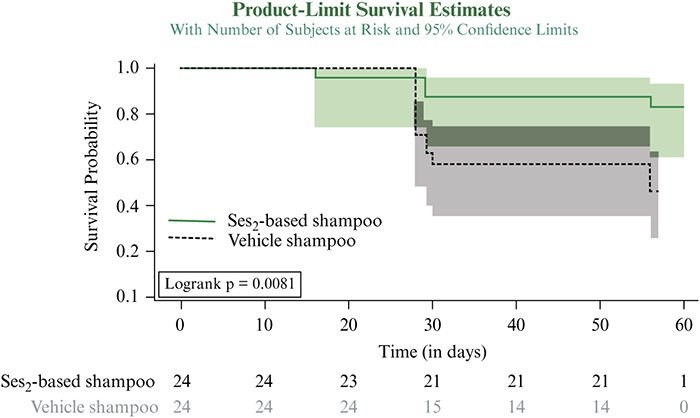
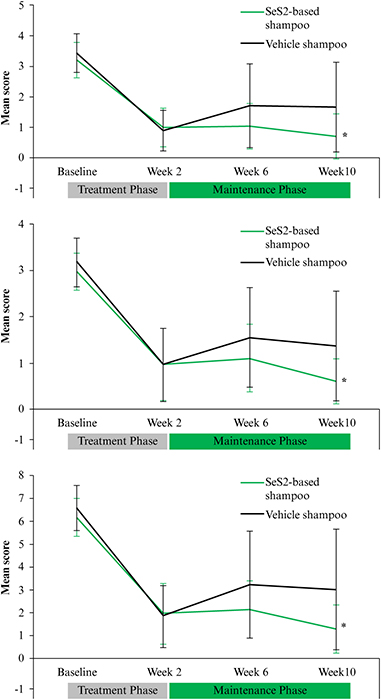
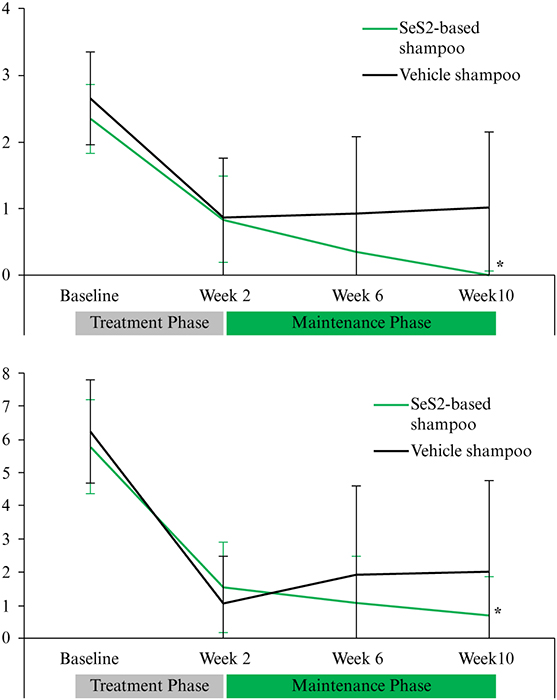
Introduction: Seborrheic dermatitis (SD) is a chronic, relapsing, inflammatory disorder of the head and trunk. Objectives: To explore the potential of a 1% Selenium disulphide (SeS2)-based shampoo to prevent relapses of scalp SD (SSD) following corticosteroid/salicylic acid (TCS/SA) treatment. Materials & Methods: After a 2-week treatment with TCS/SA, adult patients with moderate-to-severe SSD received either the SeS2-based shampoo or its vehicle for eight weeks in a randomized, double-blinded fashion. Visits took place at baseline, weeks 2, 6 and 10. SSD severity was assessed based on erythema, flakes and pruritus; patients assessed the severity of pruritus. Global investigator and patient satisfaction were assessed at week 10. Results: Forty-eight adults were included. After four and eight weeks of post TCS/SA maintenance regimen, 8.1% and 16.7% in the SeS2, and 41.7% and 54.2% in the vehicle group relapsed, respectively. First median time-to-relapse in the vehicle group was 56 days; this was not reached for SeS2. After two weeks of TCS/SA, the prevalence of patients with no pruritus was 29.2% in the SeS2 group, and 41.7% in the vehicle group; it increased to 76.2% with SeS2 and to 57.1% with the vehicle at the end of the study. The clinical benefit of treatment with TCS/SCA was maintained in the SeS2 group only. Investigators and patients were highly satisfied with the efficacy of SeS2. Tolerance to SeS2 was excellent, with no reported adverse events. Conclusion: The SeS2-based shampoo significantly reduces the time-to-relapse of moderate-to-severe SSD flares. Its tolerance was excellent, with no reported adverse events.