Hépato-Gastro & Oncologie Digestive
MENUESMO congrès (European Society For Medical Oncology) 2019: what is new in GI Oncology? Volume 26, supplement 6, Décembre 2019
- Key words: colorectal cancer, oesogastric cancer, pancreatic cancer, biliary tract carcinoma, hepatocellular carcinoma
- DOI : 10.1684/hpg.2019.1890
- Page(s) : 14-24
- Published in: 2019
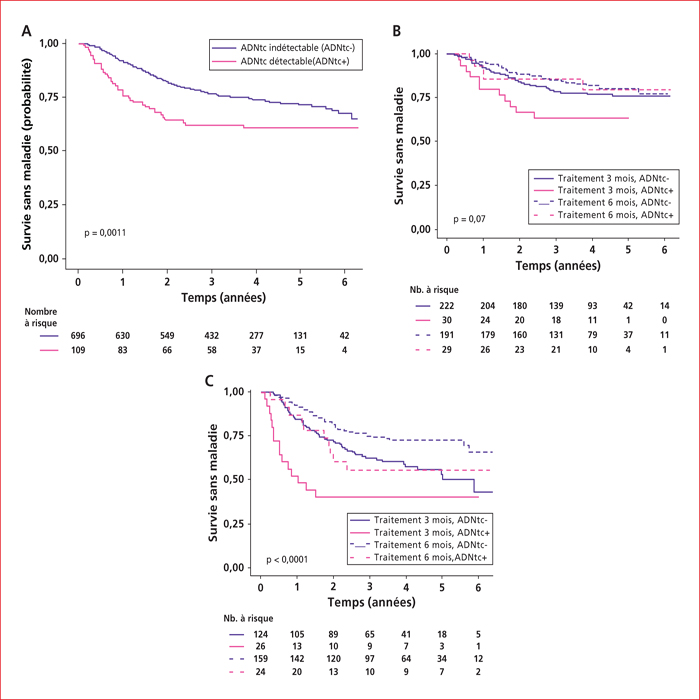
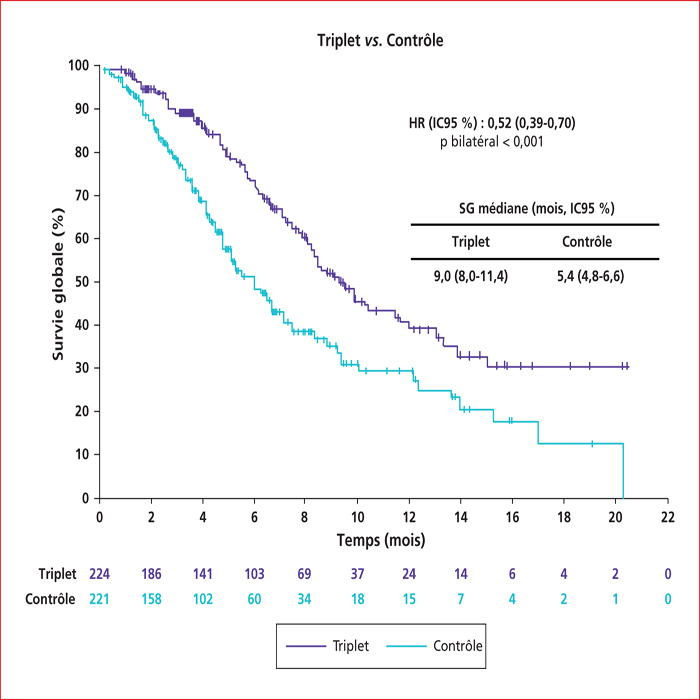
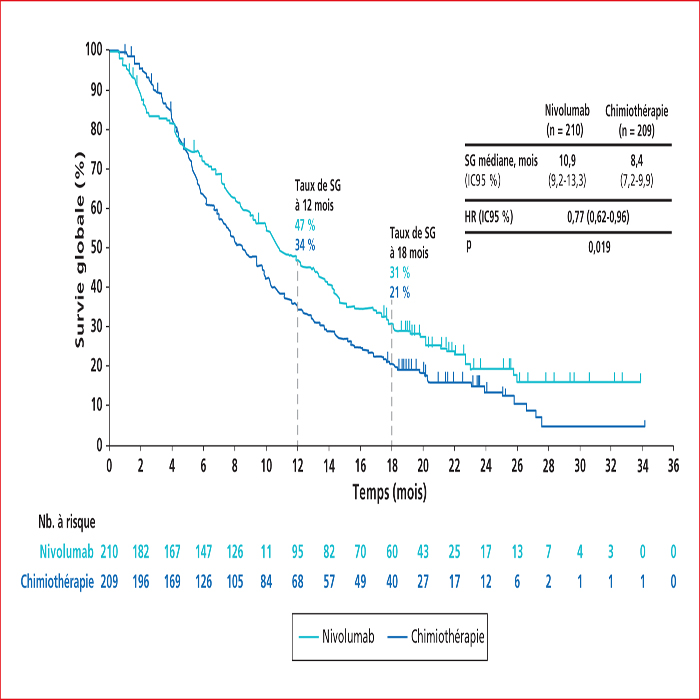
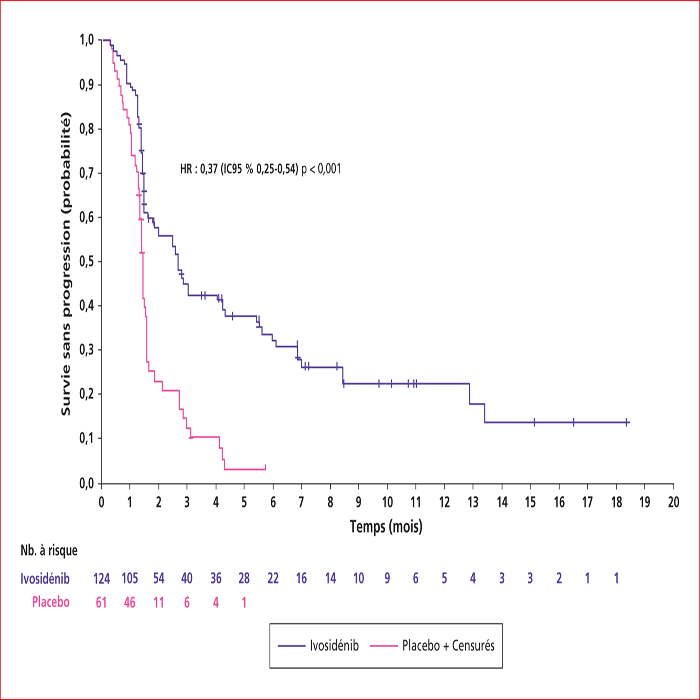
This article presents a summary of the key presentations in digestive oncology of the European Society For Medical Oncology (ESMO) conference held in Barcelona at the end of September 2019. Among them, an ancillary analysis of the IDEA France Phase III study confirmed that circulating tumor DNA is a major prognostic factor in localized colon cancers. In patients with metastatic colorectal cancer with BRAF V600E mutation, the BEACON phase III study demonstrated the superiority of the inhibition triplet encorafenib (anti-RAF)/binimetinib (anti-MEK)/cetuximab (anti-EGFR) compared to chemotherapy with FOLFIRI and cetuximab, making this triplet a new standard in the second line setting. In non-colorectal gastrointestinal cancers, nivolumab (anti-PD1) immunotherapy has been shown to be effective vs. second-line chemotherapy in advanced squamous cell carcinoma of the esophagus in the ATTRACTION-03 Phase III study. The ClarIDHy Phase III study has shown that ivosidenib, an inhibitor of the mutated form of IDH1, improves progression-free survival over placebo in patients with cholangiocarcinoma with IDH1 mutation. Ripretinib is the new fourth-line reference treatment for advanced gastrointestinal stromal tumors based on the results of the INVICTUS Phase III study, which showed superiority over placebo.
![]() This work is licensed under a
Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License
This work is licensed under a
Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License