Hématologie
MENUMultiple molecular mechanisms for chronic myeloid leukemia progression, but a possible single common origin: the genetic instability Volume 20, issue 6, Novembre-Décembre 2014
- Key words: chronic myeloid leukemia, progression, stem cell, genetic instability
- DOI : 10.1684/hma.2014.0967
- Page(s) : 308-18
- Published in: 2014
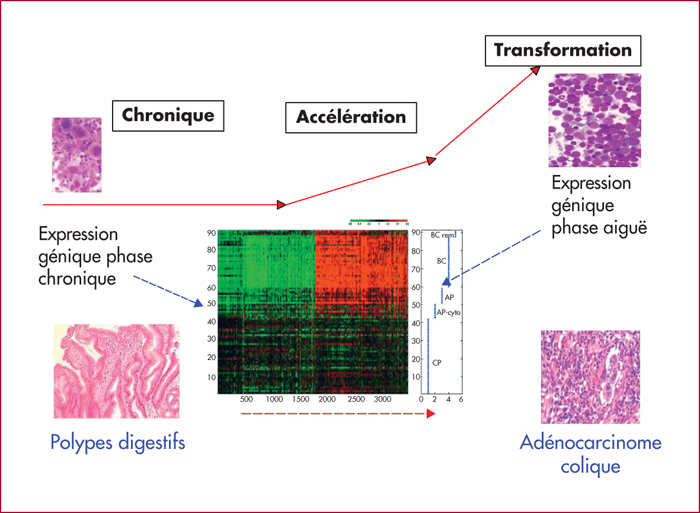
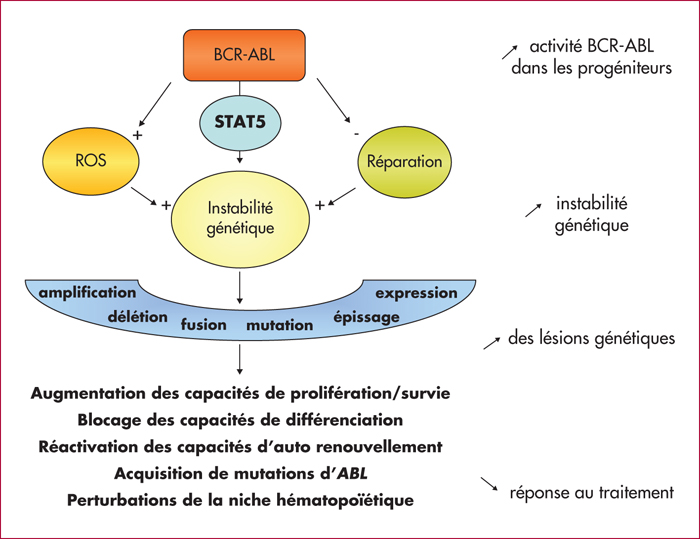
Because of its unique cytogenetic abnormality, the t(9;22)(q34;q11) translocation associated with the BCR-ABL gene fusion at molecular level, chronic myeloid leukemia (CML) is a model for studying leukemogenesis. CML progression is characterized by phenotypic variations of the myeloid progenitors, with loss of cell differentiation and apoptosis capacities and reactivation of self-renewal. Surprisingly, the three distinct phases of CML progression (chronic, accelerated, blastic) are translated at the molecular level by a biphasic gene expression profile (chronic vs accelerated and blastic phases), suggesting an irreversible mechanism. And the wide heterogeneity of acquired genetic alterations observed illustrates the complexity and the diversity of processes involved during the disease progression. However, it is supposed that this heterogeneity relies within a single origin: the genetic instability of the stem cell, presenting a BCR-ABL dependent increased production of oxygen reactive species (ROS). Moreover, BCR-ABL is supposed to reduce DNA repair efficiency, allowing the persistence of these genetic alterations. CML stem cells could be therefore the reservoir for disease progression, since these cells have been shown to lose the BCR-ABL addiction for their survival, consequently they are not eradicated by of tyrosine kinases inhibitors (TKIs).
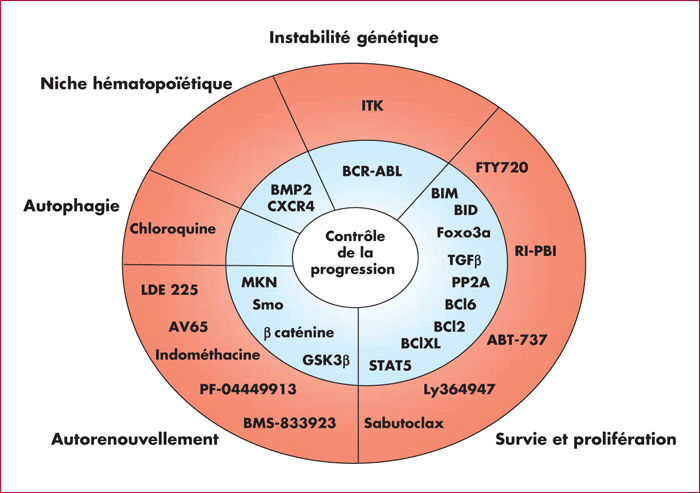
In this review, we aim to build a state of knowledge of major molecular mechanisms and affected pathways implicated during CML progression, and we discuss therapeutic options of drug associations with ITKs for the stem cell eradication and the control of genetic instability.