Hépato-Gastro & Oncologie Digestive
MENUImmunotherapy in gastroesophageal cancers Volume 29, issue 4, April 2022
- Key words: gastric adenocarcinoma, oesophageal adenocarcinoma, immunotherapy, PD-L1 expression, microsatellite instability
- DOI : 10.1684/hpg.2022.2367
- Page(s) : 514-22
- Published in: 2022
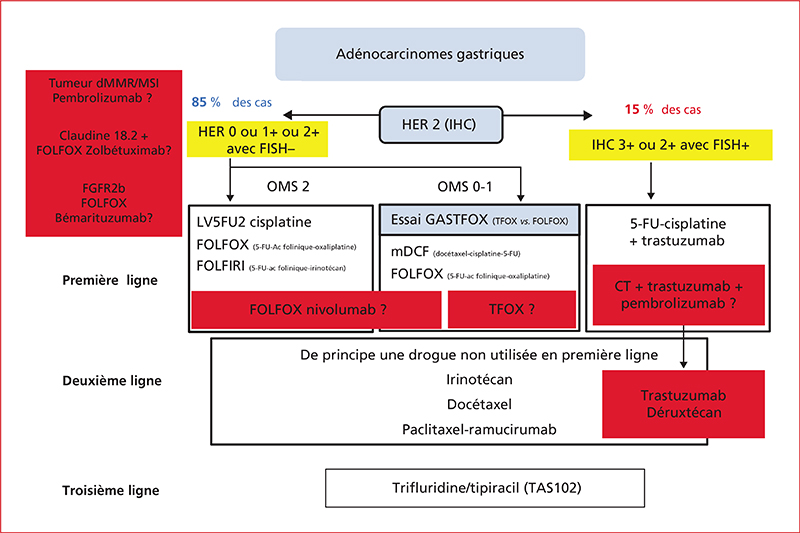
Until recently, the treatment of esophageal squamous cell carcinoma and HER-2 negative esogastric adenocarcinoma is only based on chemotherapy. Since several years, immune checkpoint inhibitors (ICI) have been evaluated in these cancers, first as monotherapy in chemo-resistant tumors and more recently in combination with chemotherapy as first-line treatment. In esophageal tumors, ICI have shown an improvement of overall survival in second line setting as monotherapy compared to chemotherapy alone, in combination with first line chemotherapy compared to chemotherapy alone in tumours with PD-L1 CPS ≥ 10 (Programmed Death Ligand-1 Combined Positive Score) and in adjuvant setting after treatment by chemoradiotherapy plus surgery compared to placebo, thus resulting in 3 European marketing authorizations. Concerning metastatic gastric adenocarcinomas with micro-satellite instability (dMMR/MSI), ICI in monotherapy (anti-PD1/PD-L1) and doublet (anti-PD1/PD-L1 plus anti-CTLA4) have shown major efficacy but reimbursement is still pending in France. Recently, fluoropyrimidine, oxaliplatin and nivolumab (anti-PD-1) combination showed its superiority over chemotherapy alone in 1st line setting in metastatic esogastric adenocarcinomas with a CPS ≥5 also resulting in a European marketing authorization. In addition, phase III trials in the perioperative setting are ongoing, combining anti-PD-1/PD-L1 with the FLOT regimen. Finally, recent trials have shown major efficacy of ICI in the perioperative setting of resectable dMMR/MSI esogastric adenocarcinoma. The major current challenge remains the identification of predictive biomarkers of ICI efficacy beyond microsatellite instability and PD-L1 expression in order to better select patients who have a survival increase with these different combinations while limiting toxicity and preserving quality of life.