Epileptic Disorders
MENUValproate-induced reversible sensorineural hearing loss: a case report with serial audiometry and pharmacokinetic modelling during a valproate rechallenge Volume 16, issue 3, September 2014
- Key words: hearing loss, ototoxicity, valproic acid, epilepsy, pharmacokinetics, AED, side effects
- DOI : 10.1684/epd.2014.0671
- Page(s) : 375-9
- Published in: 2014
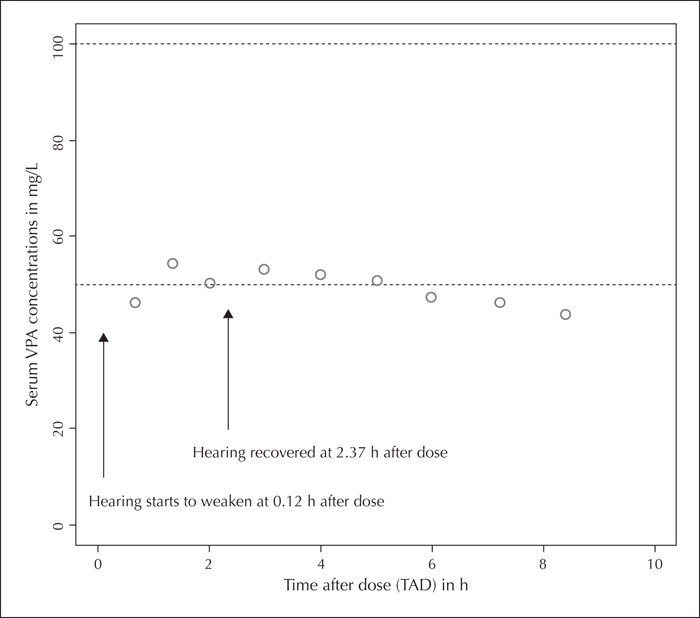
Hearing loss has been reported with valproic acid (VPA) use. However, this is the first case of VPA-induced hearing loss that was tested and confirmed with a VPA rechallenge, supported by serial audiometry and pharmacokinetic modelling. A 39-year-old truck driver with temporal lobe epilepsy was treated with VPA at 400 mg, twice daily, and developed hearing loss after each dose, but recovered within three hours. Hearing loss fully resolved after VPA discontinuation. Audiometry performed five hours after VPA rechallenge showed significant improvement in hearing thresholds. Pharmacokinetic modelling during the VPA rechallenge showed that hearing loss occurred at a level below the therapeutic range. Brainstem auditory evoked potential at three months after VPA discontinuation showed bilateral conduction defect between the cochlear and superior olivary nucleus, supporting a pre-existing auditory deficit. VPA may cause temporary hearing threshold shift. Pre-existing auditory defect may be a risk factor for VPA-induced hearing loss. Caution should be taken while prescribing VPA to patients with pre-existing auditory deficit.