Epileptic Disorders
MENUTorsional nystagmus in hypothalamic hamartoma Volume 15, issue 4, December 2013
epd.2013.0611
Auteur(s) : Aasef G Shaikh aasefshaikh@gmail.com
Department of Neurology, Case Western Reserve University, Cleveland, OH, USA
Correspondence: Aasef G Shaikh Department of Neurology, Case Western Reserve University, 11100 Euclid Avenue, Cleveland, Ohio 44106-5040, USA
Hypothalamic hamartomas are heterotopic nodules resembling normal grey matter of the hypothalamus. Generally embedded within the hypothalamus, large hamartomas may rarely extend into the pre-pontine cistern (Northfield and Russell, 1967; Balagura et al., 1979; Nishio et al., 2001). Precocious puberty, obesity, cognitive impairment, developmental delay, and gelastic seizures are the typical manifestations of hypothalamic hamartoma (Nguyen et al., 2003). Occasionally, hypothalamic hamartoma is associated with acromegaly (Scheithauer et al., 1986), hypopituitarism (Martin et al., 2003), diencephalic syndrome (Reeves and Plum, 1969), visual impairment (Valdueza et al., 1994), and episodic tonic eye deviations (Olivieri et al., 2010). This report describes the first case of pure torsional nystagmus in a patient with hypothalamic hamartoma.
Case study
A 52-year-old woman had a history of chronic epilepsy since she was 13 years old. Typical seizure semiology was tonic posturing of the right arm and gaze deviation to the right. There were fluctuating levels of consciousness and spells of mechanical laughter (gelastic seizures). The episodes were generally followed by drowsiness and lethargy. The episodes occurred once every two weeks and were refractory to multiple antiepileptics, including phosphenytoin, valproic acid, lamotrigine, oxcarbazepine, levetiracetam, lorazepam, and lacosamide. At the time of presentation, she was taking valproic acid, lamotrigine, oxcarbazepine, lorazepam, and lacosamide. Her comorbidities included hypothyroidism, hypertension, and depression.
Cognitive deficits included impairment in registering and recalling words after five minutes and simple mathematical calculations such as “serial seven”. The patient was unaware of the name of the current president of the United States and was unable to spell “world” backwards.
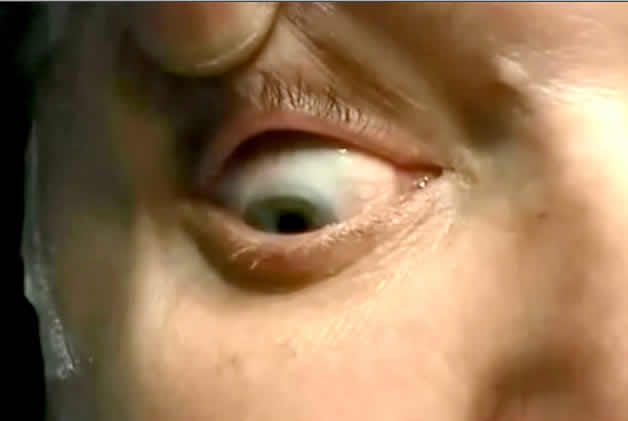
Neurological examination revealed persistent, counter clockwise beating, torsional nystagmus in straight-ahead, as well as eccentric, gaze orientation. Vertical and horizontal saccades were hypermetric but had normal velocity. Ocular pursuit, vestibulo-ocular reflex (VOR), and visual field were normal. Dysmetria was evident during reaching movements of both upper extremities, as tested by finger-to-nose manoeuvre. She was unable to walk on heels, on toes, or in tandem.
Magnetic resonance imaging revealed a mass centred within the left hypothalamus (arrows in figure 1). The mass measured 1.8 cm in mediolateral, 1.5 cm in anteroposterior, and 2.5 cm in craniocaudal dimension. The exophytic component of the mass extended into the superior pre-pontine cistern, abutting the ventral midbrain (arrows in figures 1A and 1B). Superiorly, the mass expanded into the left thalamus and partially effaced the third ventricle. Anteriorly, the mass extended to the posterior pituitary infundibulum. The mass was hyperintense on fluid attenuated inversion recovery and T2 sequences, but isointense on T1-weighted sequences. There was no contrast enhancement. The clinical and radiographic features were characteristic of hypothalamic hamartoma.
Discussion
Torsional nystagmus was the unique feature associated with hypothalamic hamartoma in our patient. Mesencephalic lesion, possibly secondary to mechanical compression from the hypothalamic hamartoma that extended into the pre-pontine cistern, could account for torsional nystagmus. Two vital midbrain nuclei, in the vicinity of observed compression from hamartomas, are responsible for torsional and vertical eye movements: the rostral interstitial nucleus of medial longitudinal fasciculus (riMLF) and the interstitial nucleus of Cajal (INC) (Villis et al., 1989; Crawford et al., 1991). The riMLF is responsible for generation of torsional-vertical saccades (Villis et al., 1989), while the INC is a torsional-vertical neural integrator that holds the eyes steady in a desired gaze orientation (Crawford et al., 1991). In animal models, isolated experimental lesions of riMLF result in contra-lesional tonic torsional deviation, contra-lesional beating torsional nystagmus, loss of ipsi-lesional torsional saccades, decreased reactivity of torsional and vertical VOR, and decreased velocity of torsional and vertical saccades (Helmchen et al., 1998; Helmchen and Buttner, 2000; Rambold et al., 2000). In contrast, an isolated lesion of the INC leads to contra-lesional torsional deviations, followed by ipsi-lesional torsional quickphases, but normal velocities of torsional and vertical saccades and normal reactivity of torsional and vertical VOR (Helmchen et al., 1998). Given the proximity of riMLF and INC, even small lesions can cause their combined dysfunction, sometimes associated with additional phenomenology, such as vertical gaze evoked nystagmus and seesaw nystagmus (Halmagyi et al., 1994; Helmchen et al., 2002). In our patient, the torsional nystagmus was beating counter clockwise, hence, the quickphases were directed to the left side; ipsilateral to the location of hypothalamic hamartoma, abutting the mesencephalon. Therefore, in this patient, torsional nystagmus can be explained by INC dysfunction due to compression from the hypothalamic hamartoma.
Although torsional nystagmus is prominent in subjects with unilateral INC lesions, vertical nystagmus with variable severity may be seen. In our patient, vertical nystagmus, however, could not be appreciated during clinical examination. The laterality of torsional nystagmus and normal velocity of vertical saccades suggested that function of riMLF was spared in our patient.
Our patient had medically refractive epilepsy and was exposed to multiple antiepileptics, including phosphenytoin and valproic acid. These antiepileptics can lead to reversible cerebellar dysfunction; rarely, phosphenytoin can even cause irreversible injury to the Purkinje neurons (Horne, 1973; McLain et al., 1980). It is possible that dysmetria during reaching hand movements and the heel-shin manoeuvre could be related to chronic epilepsy or antiepileptics (Horne, 1973; Hagemann et al., 2002).
In conclusion, an ipsilaterally beating torsional nystamus was observed in a patient with large hypothalamic hamartoma. Compressive lesion of the midbrain interstitial nucleus of Cajal is the speculated cause of torsional nystagmus.
Disclosures
The author has no conflicts of interests to disclose.
Legends for video sequences Ipsi-lesionally beating torsional nystagmus in a patient with hypothalamic hamartoma Key words for video research on www.epilepticdisorders.com Syndrome: gelastic seizures with hypothalamic hamartoma Etiology: hamartoma (hypothalamic) Phenomenology: gelastic seizure; oculoclonic seizures; nystagmus Localization: hypothalamus