Epileptic Disorders
MENUA systematic review of the efficacy of perampanel as treatment for myoclonic seizures and symptomatic myoclonus Volume 24, issue 4, August 2022
- Key words: perampanel, myoclonic seizures, progressive myoclonus epilepsy, idiopathic generalized epilepsy, myoclonus
- DOI : 10.1684/epd.2022.1439
- Page(s) : 633-46
- Published in: 2022
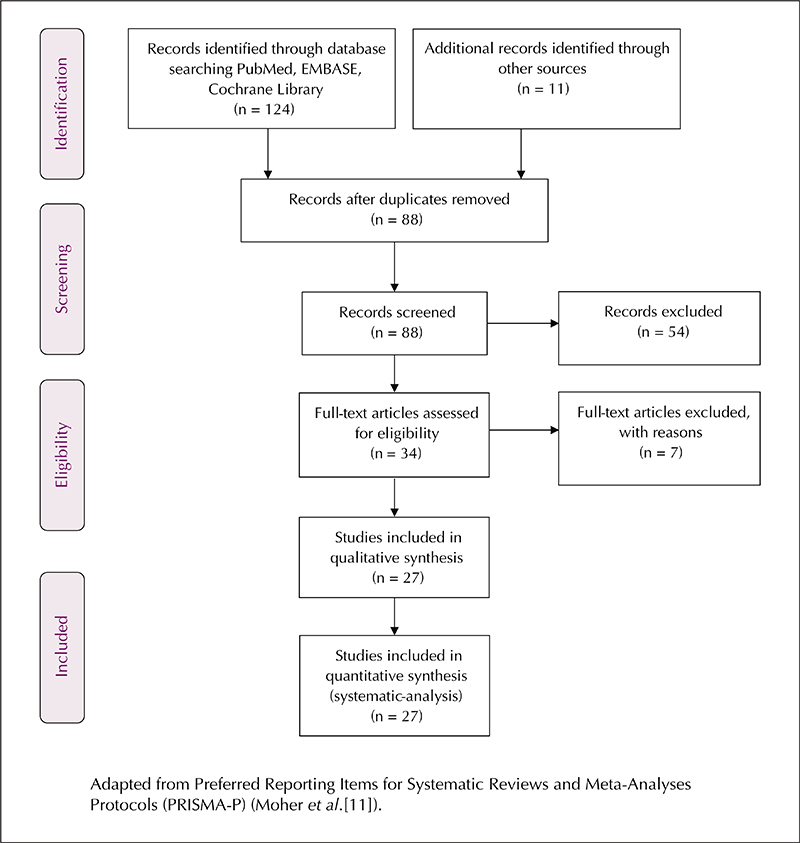
Epileptic myoclonus or myoclonic seizures can occur in idiopathic generalized epilepsy (IGE) and progressive myoclonus epilepsy (PME). However, symptomatic myoclonus which is stimulus-sensitive and provoked by movement is typically seen in PME and Lance-Adams syndrome. Symptomatic myoclonus is not always associated with epileptiform discharges on the electroencephalogram. Therapeutic interventions such as anti-seizure medications (ASMs), the ketogenic diet and vagus nerve stimulation are not always effective. There is emerging evidence that perampanel (PER), an α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid (AMPA) receptor antagonist, may be effective for the treatment of myoclonic seizures and symptomatic myoclonus. We performed a systematic review of the literature to assess the efficacy of PER as treatment for myoclonic seizures and symptomatic myoclonus. Twenty-seven studies with a total sample size of 260 patients were included. The efficacy of PER was analysed separately for myoclonic seizures and symptomatic myoclonus. In the group with myoclonic seizures, 50% responder, 75% responder and seizure freedom rates were reported as 74.3% (101/136), 60.3% (82/136) and 57.4% (78/136), respectively, with a follow-up duration of 6-12 months. However, in one post-hoc analysis of data from patients with IGE, the efficacy of PER as treatment for myoclonic seizures during the double-blind phase showed no significant difference compared to placebo. The efficacy of PER for symptomatic myoclonus was reported in a total of 119 patients. Four studies (n=88 patients) reported the efficacy of PER as a decrease in myoclonus score/scale. In the remaining 31 patients, symptomatic myoclonus resolved in three patients, decreased in 21 patients and seven patients showed no improvement. We also analysed the number of patients who were already on levetiracetam (LEV) or valproic acid (VPA) at the time of PER initiation; these data were available for 153 patients. Of these, 56.8% were on LEV and 75.1% were on VPA when PER was initiated. This systematic review suggests that PER may be effective as treatment for drug-resistant myoclonic seizures and symptomatic myoclonus. It may also be effective in patients who have already failed to respond to LEV and VPA. These findings are preliminary yet encouraging. This study has several limitations, particularly given the scarcity of high-quality randomized controlled trials and marked heterogeneity regarding the type and results of the studies. Hence, the findings of this review should be viewed with considerable reservation.