Epileptic Disorders
MENUA child with ictal vocalizations and generalized epilepsy Volume 17, issue 1, March 2015
Ictal vocalizations may take numerous forms; both articulate speech and non-speech vocalizations have been described in focal epilepsies, with seizures mainly originating from the frontal and temporal lobe (Janszky et al., 2000; Horvath et al., 2009). Non-speech productions include peculiar sounds, such as barking or humming (Bartolomei et al., 2002 ; Patra et al., 2011). To the best of our knowledge, this phenomenon has not been described in generalized epilepsies. We present the case of an adolescent boy with juvenile-onset generalized epilepsy who presented with ictal “ovine vocalizations”.
Case study
This previously healthy 13-year-old boy presented to our neurology clinic after a tonic-clonic seizure which was reported to be generalized from onset, while he was watching television. Detailed patient history revealed that, for the preceding four months, he had presented with paroxysmal episodes of vocalizations resembling the bleating of a sheep, accompanied by a brief twitching of the mouth, lasting a few seconds, sometimes associated with jerking of the upper limbs. No focal clonias, respiratory sounds, or version of the eyes or head were noted, he did not feel anything during these attacks, there was no aura before, and he was totally amnesic to the seizures. These episodes were initially considered as tics by the parents, but rapidly became a source of embarrassment to their son, especially at school. His neurological examination was normal. Family history was positive for epilepsy; his mother was treated for repeated seizures of imprecise semiology in childhood but is currently seizure-free.
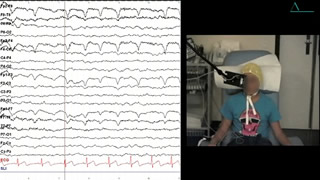
Routine EEG revealed a normal 10-Hz background rhythm (figure 1), and photosensitivity at 60 Hz with episodes of vocalizations as described above, correlating with time-locked discharges of generalized spikes and polyspikes (see video-EEG sequence) (60-Hz, high-frequency stimulation is used in our lab for all patients presenting with generalized seizures; epileptiform discharges are only seen in patients who have epilepsy with photosensitivity). The child was examined during the video-EEG recording and there was complete loss of consciousness during these brief seizures. Video-EEG monitoring over 24 hours revealed several bursts of 3-Hz, bilateral spike-and-wave discharges lasting for 5-12 seconds, sometimes accompanied by vocalizations, but sometimes without any clinical manifestations (figure 2).
High-resolution 3 T brain MRI was normal and, in particular, did not show any structural lesion in the frontal or temporal lobes. The boy was treated with levetiracetam with unfavourable clinical response, despite increasing doses up to 50 mg/kg/day. Due to the persistence of both types of seizures, the treatment was changed to valproate, to which he responded rapidly, without recurrence of any seizure type after a follow-up of 12 months, at a daily dose of 20 mg/kg. The blood levels of valproate tested after four weeks were 456 μmol/l (reference values being 350-700 μmol/l).
Discussion
Ictal vocalizations have been mostly described in the literature in relation to hemispheric lateralization of the epileptogenic zone. In a previous study, ictal vocalizations were defined as audible sounds without speech quality, that did not accompany apnoea, GTC seizures or clonic seizures, and were an obvious ictal phenomenon (Janszky et al., 2000). In this report, ictal non-speech vocalizations and focus lateralization were studied by video-EEG monitoring in 27 patients. Pure ictal vocalizations occurred in 11 patients, nine of whom had a left frontal epileptogenic zone. It was proposed that pure ictal vocalizations indicated a left hemispheric seizure onset and implicated epileptic activity involving the Broca area or the supplementary sensorimotor area (Janszky et al., 2000). On the contrary, pure ictal vocalizations were reported to occur significantly more frequently during seizures with right hemispheric onset, thus contradicting the suggestion of seizure origin in the speech-dominant hemisphere, although this symptom showed no lateralizing significance in individual patients (Bonelli et al., 2007).
Further, isolated ictal vocalizations were described as the only observable clinical manifestation of left frontal lobe epilepsy in a case report by Rego et al. (2006). Ictal vocalizations have also been described to be associated with epilepsy originating from the left temporal lobe (Horvath et al., 2009). In the latter report, the authors retrospectively studied patients with surgically-treated TLE and seizure freedom. Before surgical intervention, all patients had ictal vocalizations during focal dyscognitive seizures, although none of the seizures occurred with exclusively short episodes of vocalization. It should be noted that simultaneous EEG recordings were not reported.
Ictal speech manifestations are classified as vocalizations, normal speech or abnormal speech (Gabr et al., 1989). In their semiological study of intractable focal seizures, Gabr et al. reported that ictal vocalization occurred in 48.5% of their 35 patients, while ictal normal (identifiable) speech occurred in 34.2% of patients. Abnormal speech (arrest, dysphasia, dysarthria, or use of non-identifiable words) occurred in 51.4% of patients, either ictally or postictally. Of all these manifestations, only postictal dysphasia and ictal identifiable speech had significant lateralizing value; 92% of patients with postictal dysphasia had seizures originating from the dominant temporal lobe (p<0.001), and 83% of those with ictal identifiable speech had onset in the non-dominant side (p=0.013). The authors observed that patients with seizures originating from the dominant hemisphere had more frequent ictal vocalizations than those with non-dominant foci (Gabr et al., 1989). Koerner et al. (1988) similarly reported that in 84 patients with focal dyscognitive seizures evaluated by EEG and video telemetry, who went on to have epilepsy surgery, complex intraictal vocalizations and postictal language dysfunction were found to have strong lateralizing significance.
Disordered non-verbal expressions include non-speech vocalizations, verbal automatisms or jargon. Ictal barking has been described as a manifestation of mesial frontal epilepsy (Patra et al., 2011) or of seizures originating from the temporal lobe (Harandi et al., 2012). Patra et al. described a 42-year-old, right-handed male with post-traumatic, drug-resistant complex partial epilepsy who manifested with ictal barking close to electrographic onset. Extraoperative electrocorticography with subdural electrode coverage of the right frontoparietal, right temporal and left frontal surfaces provided the identification of an ictal origin in the right mesial temporal lobe with barking vocalization manifesting within three seconds of electrographic onset. There was no subsequent spread of activity noted beyond the temporal lobe. Resection of the mesial temporal structure resulted in seizure freedom (Patra et al., 2011). Finally, ictal humming was reported in three patients with seizures originating from the temporal lobe; in two on the left and in one on the right side (Bartolomei et al., 2002). Ictal humming has been reported as automatisms in absence seizures in two patients (Laoprasert, 1999).
To the best of our knowledge, ictal ovine vocalizations (sheep bleating sounds) have never been described in relation to any type of epilepsy. We assume that vocalization during tonic or myoclonic seizures is the result of the involuntary contraction of the diaphragmatic and laryngeal muscles and consider this as the most reasonable explanation for this ictal phenomenon in our patient. The generalized ictal correlate, normal neurological examination and cerebral MRI, and complete control of seizures with low doses of valproic acid support the notion that these vocalizations are the result of generalized epilepsy.
We conclude that ictal non-speech vocalizations may be observed not only in focal or structural epilepsies, as is the current belief, but also in generalized epilepsies, however, the exact underlying mechanism of this ictal phenomenon needs to be further delineated.
Acknowledgements and disclosures
We thank Dr L. Spinelli for the preparation of the video-EEG images and Dr A. Fogarasi for sharing his semiological expertise with us. This work was not supported by any grant.
None of the authors have any conflict of interest to disclose.