European Journal of Dermatology
MENUPimecrolimus 1% cream for mild-to-moderate atopic dermatitis: a systematic review and meta-analysis with a focus on children and sensitive skin areas Volume 33, issue 5, September-October 2023
Figures
Tables
- Key words: atopic dermatitis, pimecrolimus, tacrolimus, crisaborole, topical corticosteroids, meta-analysis
- DOI : 10.1684/ejd.2023.4556
- Page(s) : 474-86
- Published in: 2023
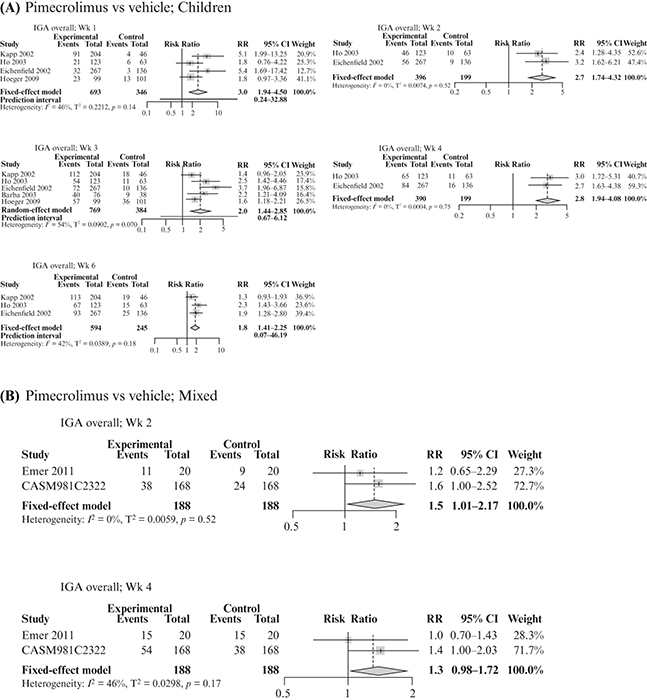
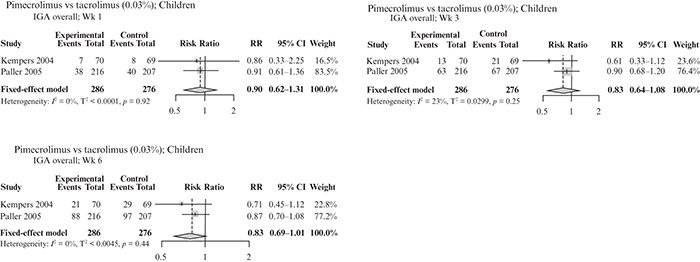
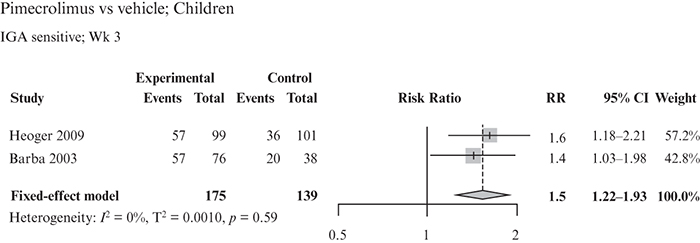
This systematic literature review (SLR) and meta-analysis assessed the efficacy and safety of pimecrolimus vs other topical treatments in patients with mild-to-moderate atopic dermatitis (AD), focusing on children and sensitive skin areas. An SLR was conducted in MEDLINE, Embase and Cochrane Library databases on January 15th, 2020, to identify randomized controlled trials (RCTs) with pimecrolimus as a study arm. Another SLR performed on October 5th, 2020 identified RCTs with a crisaborole study arm. Direct pair-wise meta-analysis was used to compare pimecrolimus with vehicle, tacrolimus or topical corticosteroids (TCS; n = 27 studies). Outcomes included Investigator’s Global Assessment (IGA) score 0/1 up to week 6 and adverse events. Pimecrolimus was more efficacious than vehicle in achieving IGA 0/1 up to week 6 in children, and similar safety profiles were observed with pimecrolimus and vehicle in children and the mixed population, including on sensitive skin. No significant differences in efficacy and safety were observed between pimecrolimus and tacrolimus 0.03%. Efficacy and safety were similar for pimecrolimus and mild medium potency TCS; mildly potent steroids caused transient epidermal thinning in sensitive skin areas (not seen with pimecrolimus). Pimecrolimus can be considered as a first-line option for mild-to-moderate AD, particularly in children and sensitive skin areas.
![]() This work is licensed under a
Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License
This work is licensed under a
Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License