Epileptic Disorders
MENUA comprehensive clinico-pathological and genetic evaluation of bottom-of-sulcus focal cortical dysplasia in patients with difficult-to-localize focal epilepsy Volume 21, numéro 1, February 2019
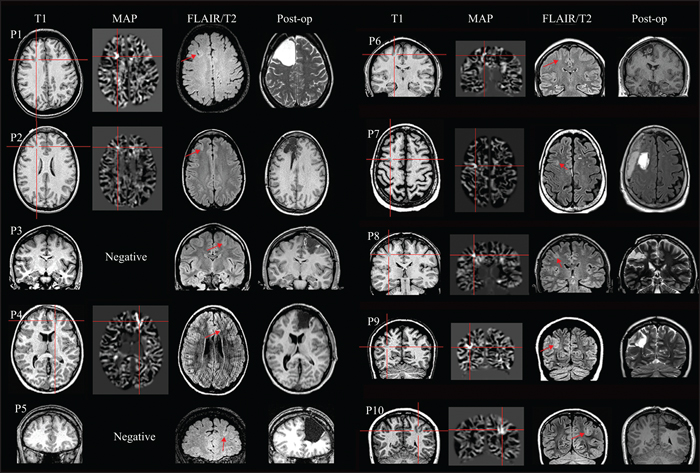
Figure 1
MRI findings and post-processing results for all patients included in the study. The first two columns are the preoperative T1w MPRAGE images and the coregistered MAP grey-white junction output. The crosshair pinpoints the location of the lesion. Absence of the MAP junction image indicates that the MAP processing was negative. The third column is the preoperative FLAIR/T2 images, whichever best depict the lesion (shown by arrow) in that particular patient. The rightmost column shows the postoperative MRI, indicating the extent of resection of the lesions.
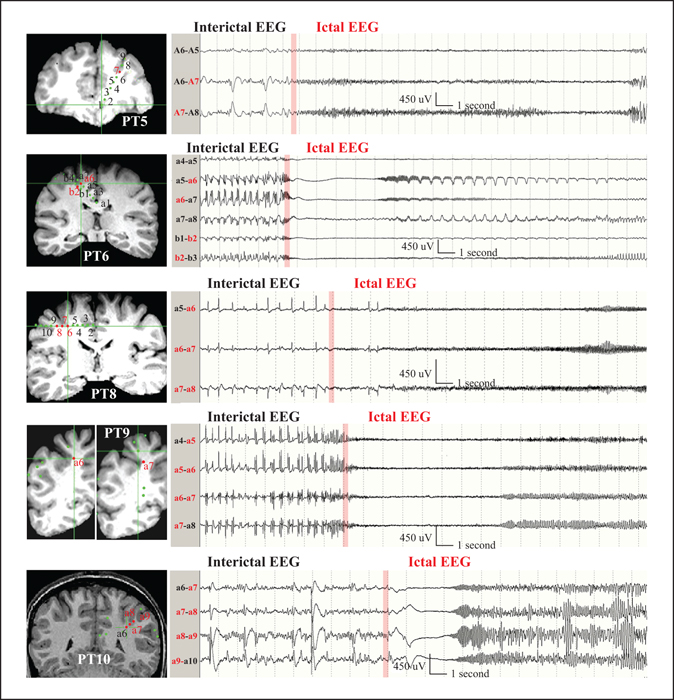
Figure 2
Location of the depth electrode contacts and their anatomic relation to BOS lesions as co-registered on T1-weighted MRI coronal cuts in five patients are shown on the left. The electrode contacts recording ictal EEG onset are labelled in red. The samples of EEG channels recording ictal EEG onset from the depth electrodes (bipolar montage) are shown on the right. The red bars point to the ictal EEG onsets that were preceded by the preictal repetitive spikes/polyspikes in each of the patients. Patients (PT) 6 and 9 showed interictal rhythmic polyspikes and wave discharges that became more frequent immediately prior to the ictal onset. Patients 5 and 10 showed interictal repetitive spikes that were intermixed with low-voltage fast activities. In all patients, ictal EEGs showed tonic fast-frequency discharges. Seizures in Patients 6 and 10 were characterized by a brief initial attenuation of the EEG prior to the emergence of the low-amplitude fast activities.
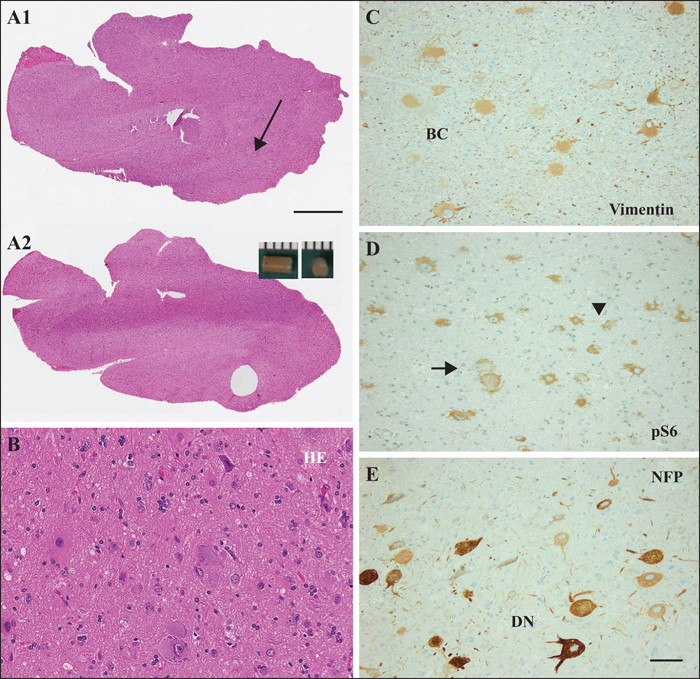
Figure 3
(A-C) H&E staining: whole-slide imaging of Patient 9 (table 1) before (A1) and after (A2) microdissection with a 2-mm diameter punching device (scale bar = 4 mm); (B) higher magnification of area indicated by arrow in (A) and used for DNA extraction following micro-dissection (shown in [A’]). (C) Vimentin immunohistochemistry highlighting balloon cells (BC); serial section from (B). (D) phosphorylated S6 (pS6) immunohistochemistry highlighting both FCD II cell types, dysmorphic neurons (arrow), and balloon cells (arrowheads); serial section from (B). (E) Immunohistochemistry for neurofilament protein highlighting dysmorphic neurons (DN); serial section from (B). Microscopic measurements reveal 8% of cells with a FCDII phenotype. Scale bar in (B-E) = 50 μm.
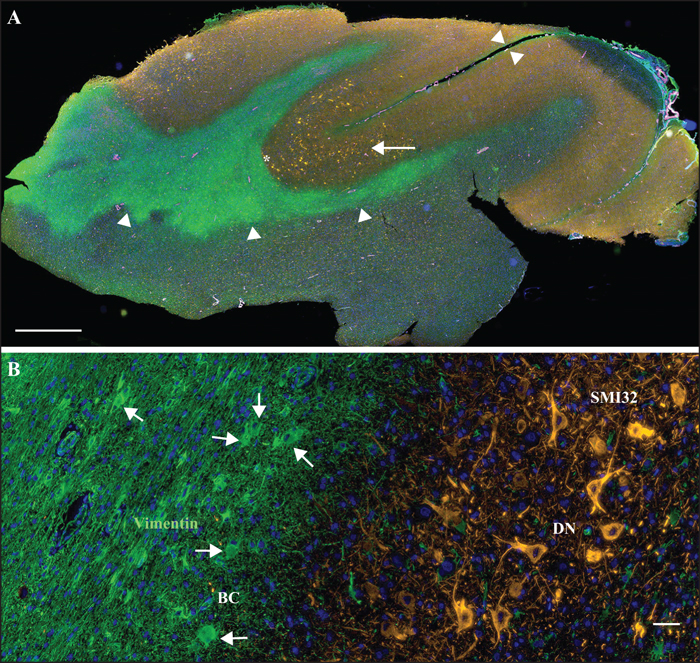
Figure 4
Surgical histopathology of Patient 9 (table 1). (A) Concentration of dysmorphic neurons (arrow) decorated with anti-non-phosphorylated neurofilament H-specific antibodies (clone SMI32, Alexa555-labeled anti-mouse IgG1 secondary antibody, orange pseudocolour) at the bottom of a sulcus (sulcal surface indicated by small arrowheads), with concomitant accumulation of vimentin-positive balloon cells (clone SP20, Alexa488-labeled anti-rabbit IgG secondary antibody, green pseudocolour; arrowheads) in the underlying white matter. In addition, vascular myocytes expressing smooth muscle actin (clone 1A4, Alexa647-labelled anti-murine IgG2a secondary antibody, magenta pseudocolour) and nuclei (Hoechst 33342, blue pseudocolour) are visualized (multichannel-immunofluorescence whole slide imaging, 3DHistech MIDI). (B) High-power magnification of area indicated by asterisk in (A) showing predominant localization of vimentin-immunopositive balloon cells (BC) in white matter (as indicated by arrows) and SMI32-immunopositive dysmorphic neurons (DN) in grey matter. Scale bar in (A) = 2 mm, in (B) = 50 μm.

