Magnesium Research
MENUIs serum magnesium level associated with atrial fibrillation in the mixed medical/surgical intensive care unit setting? Volume 35, numéro 3, 2022-07-01
- Mots-clés : atrial fibrillation, critical care, hospital mortality, intensive care unit, magnesium
- DOI : 10.1684/mrh.2023.0506
- Page(s) : 96-107
- Année de parution : 2022
Background
Although low serum magnesium level is a a relatively common problem in mixed medical/surgical intensive care units (ICUs), its association with new-onset atrial fibrillation (NOAF) has been studied to a lesser extent. We aimed to investigate the effect of magnesium levels on the development of NOAF in critically ill patients admitted to the mixed medical/surgical ICU.
Methods
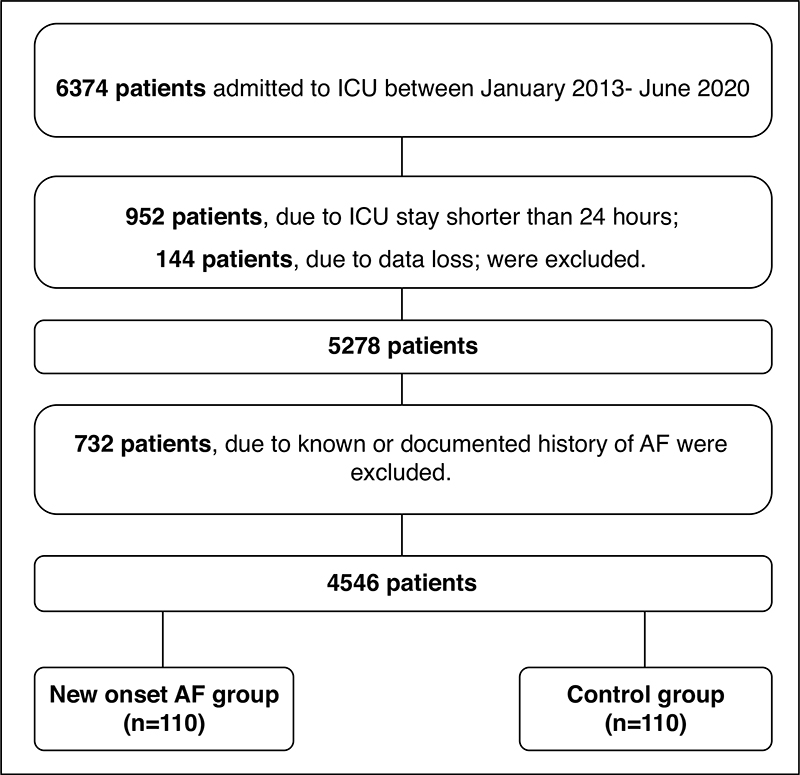
A total of 110 eligible patients (45 female, 65 male) were included in this case-control study. The age and sex-matched control group (n = 110) included patients with no atrial fibrillation from admission to discharge or death.
Results
The incidence of NOAF was 2.4% (n = 110) between January 2013 and June 2020. At NOAF onset or the matched time point, median serum magnesium levels were lower in the NOAF group than in the control group (0.84 [0.73-0.93] vs. 0.86 [0.79-0.97] mmol/L; p = 0.025). At NOAF onset or the matched time point, 24.5% (n = 27) in the NOAF group and 12.7% (n = 14) in the control group had hypomagnesemia (p = 0.037). Based on Model 1, multivariable analysis demonstrated magnesium level at NOAF onset or the matched time point (OR: 0.07; 95%CI: 0.01-0.44; p = 0.004), acute kidney injury (OR: 1.88; 95%CI: 1.03-3.40; p = 0.039), and APACHE II (OR: 1.04; 95% CI: 1.01-1.09; p = 0.046) as factors independently associated with an increased risk of NOAF. Based on Model 2, multivariable analysis demonstrated hypomagnesemia at NOAF onset or the matched time point (OR: 2.52; 95% CI: 1.19-5.36; p = 0.016) and APACHE II (OR: 1.04; 95%CI: 1.01-1.09; p = 0.043) as factors independently associated with an increased risk of NOAF. In multivariate analysis for hospital mortality, NOAF was an independent risk factor for hospital mortality (OR: 3.22; 95% CI: 1.69-6.13, p<0.001).
Conclusion
The development of NOAF in critically ill patients increases mortality. Critically ill patients with hypermagnesemia should be carefully evaluated for risk of NOAF.