Epileptic Disorders
MENUIncRNA UCA1 induces autophagic gene expression via epigenetic regulation mediated by ATG16L1 and miR-132-3p in SH-SY5Y cells treated with retinoic acid Volume 24, numéro 5, October 2022
- Mots-clés : lncRNA UCA1, ATG16L1, miR-132-3p, epilepsy, autophagy
- DOI : 10.1684/epd.2022.1477
- Page(s) : 917-27
- Année de parution : 2022
Objective
Epilepsy is a chronic brain disease with recurrent seizures. Autophagy plays a crucial role in the progression of epilepsy. This study aimed to explore the function and intrinsic mechanism of the long non-coding RNA (lncRNA) UCA1/miR-132-3p/ATG16L1 axis in epilepsy via regulation of autophagy.
Methods
The expression of lncRNA UCA1, miR-132-3p and ATG16L1 was measured in serum from epileptic patients by quantitative RT-PCR. A SH-SY5Y cell model was further constructed using retinoic acid to investigate the UCA1/miR-132-3p/ATG16L1 axis by quantitative RT-PCR, western blotting, fluorescence in situ hybridisation, RNA immunoprecipitation, chromatin immunoprecipitation, and a dual-luciferase reporter gene assay.
Results
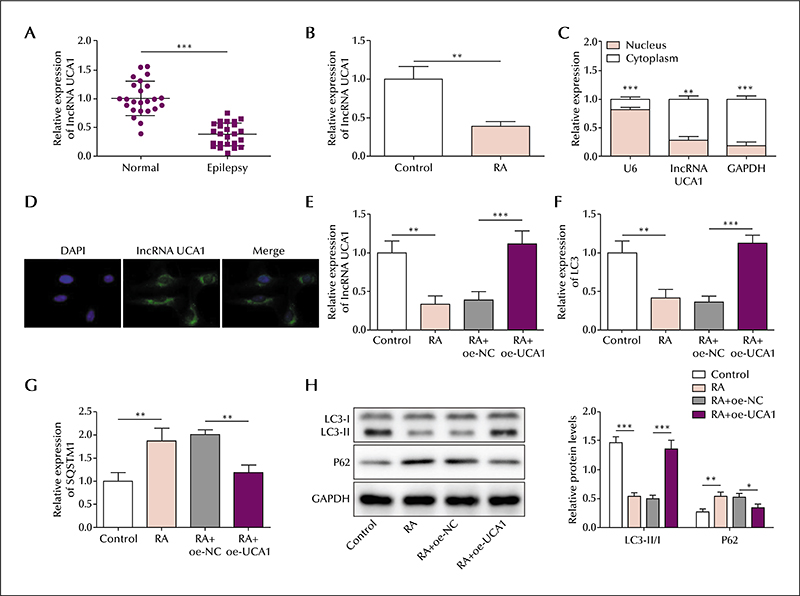
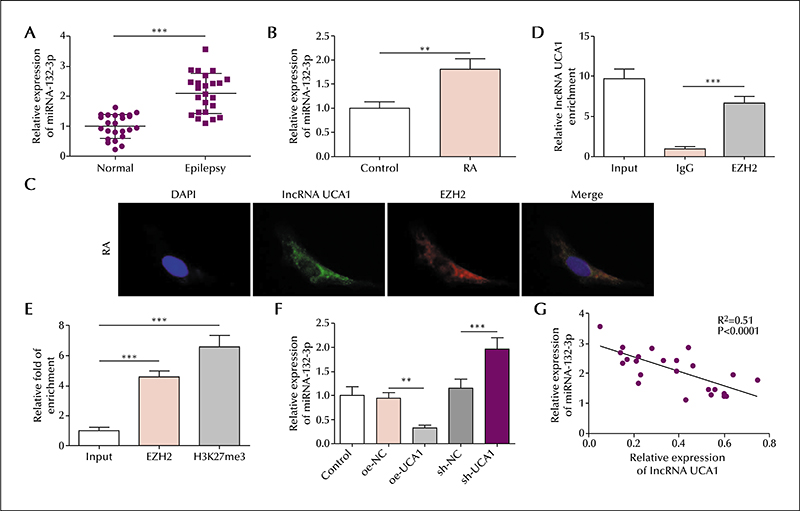
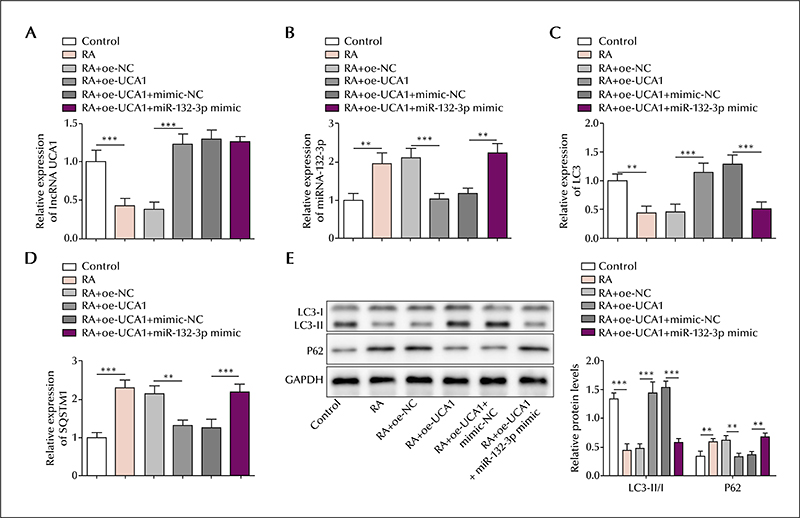
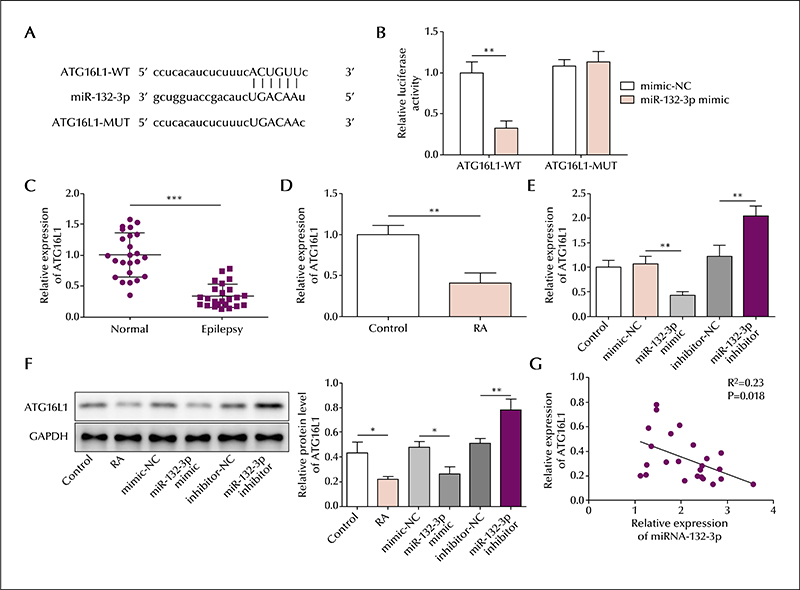
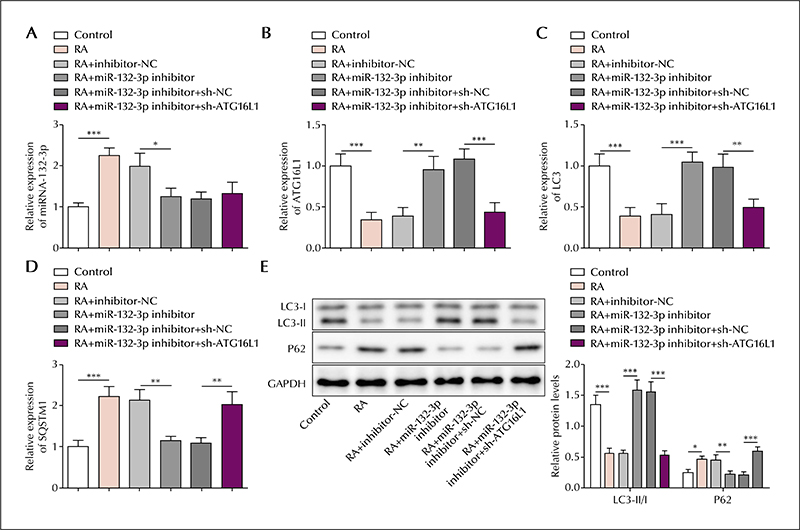
In the serum of epileptic patients, the level of lncRNA UCA1 and ATG16L1 was reduced and miR-132-3p elevated, compared to controls. Similarly, in the SH-SY5Y cell model, the level of lncRNA UCA1 and ATG16L1 was reduced and miR-132-3p elevated in retinoic acid-treated cells; lncRNA UCA1 was mainly located in the cytoplasm. lncRNA UCA1 overexpression was shown to promote autophagic gene expression, which was reversed by miR-132-3p overexpression. Moreover, autophagic gene expression induced by miR-132-3p knockdown was reversed by ATG16L1 knockdown. Based on precipitation assays, lncRNA UCA1 and miR-132-3p were shown to form a complex with the transcription factor, EZH2, and miR-132-3p was shown to interact with ATG16L1 based on a luciferase assay. Finally, lncRNA UCA1 was shown to negatively regulate miR-132-3p expression, and miR-132-3p was shown to negatively regulate ATG16L1.
Significance
In this cell model, lncRNA UCA1 promotes autophagic gene expression via epigenetic regulation mediated by ATG16L1 and miR-132-3p.