Epileptic Disorders
MENUEvaluation of real-world effectiveness of perampanel in Japanese adults and older adults with epilepsy Volume 24, numéro 1, February 2022
- Mots-clés : perampanel, aged epilepsy patients, Japan, safety, effectiveness
- DOI : 10.1684/epd.2021.1369
- Page(s) : 123-32
- Année de parution : 2022
Objective. While previous studies have demonstrated the safety and effectiveness of perampanel (PER) in combination with other anti-seizure medications in adult patients, data for older patients are limited. This study aimed to confirm real-world safety and effectiveness of combination treatment with PER in Japanese patients with focal seizures with or without focal to bilateral tonic-clonic seizures (FBTCS) or generalised tonic-clonic seizures (GTCS) according to age subgroups (
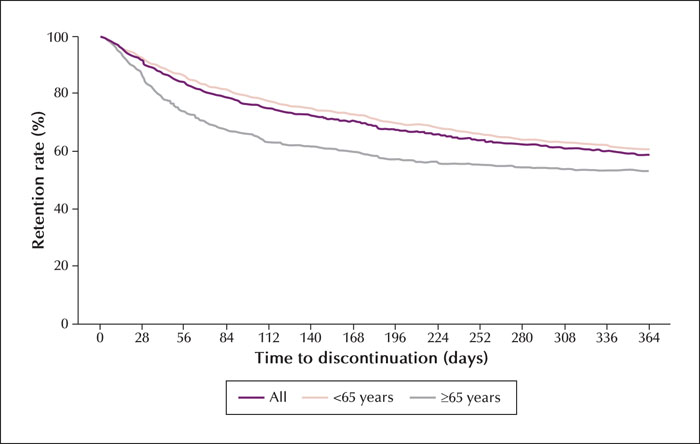
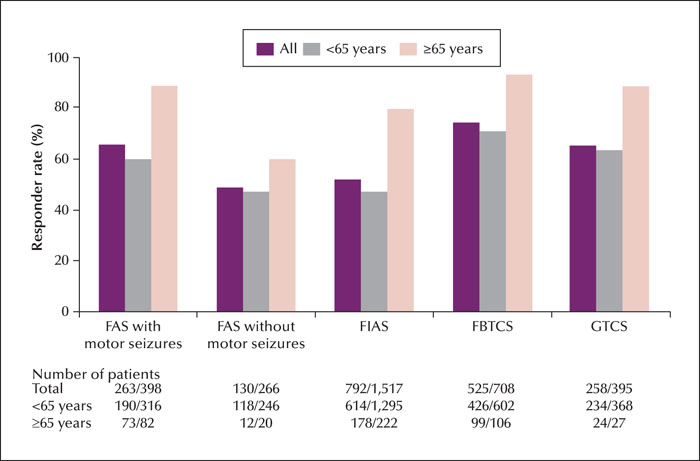
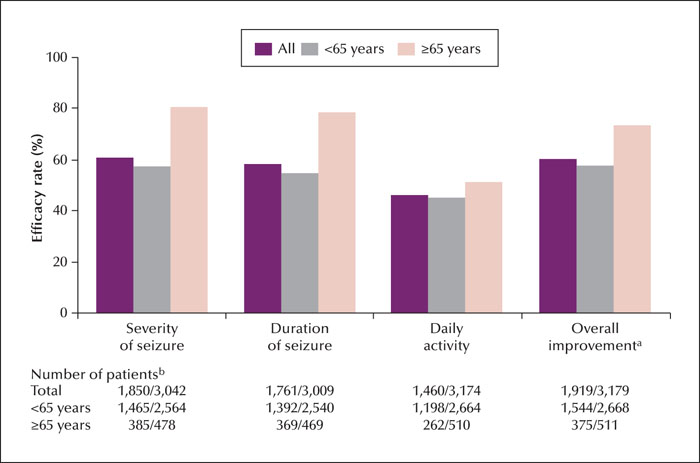
Methods. This large-sample prospective post-marketing observational study included a 24-52-week observation period after the first PER treatment. Safety was assessed according to adverse drug reactions (ADRs) and efficacy was evaluated based on the 50% responder rate and rates of overall symptom improvement.
Results. Among the 3,808 patients who were enrolled, 3,716 (3,026 patients aged
Significance. PER was effective in reducing seizure frequency and was safe, especially in older patients. PER may be a clinical treatment option for older patients with seizure disorders.