European Journal of Dermatology
MENUSafety and efficacy of secukinumab in psoriasis patients infected with hepatitis B virus: a retrospective study Volume 32, numéro 3, May-June 2022
Psoriasis is a type of immune-mediated inflammatory skin disorder that affects up to 3% of the population worldwide [1]. Psoriasis can be difficult to treat because it is accompanied frequently by various comorbidities.
Secukinumab is a fully human immunoglobulin-G1-kappa monoclonal antibody targeting interleukin (IL)-17A. Secukinumab has demonstrated high efficacy for the treatment of plaque psoriasis. In 2015, it was the first (and only) human IL-17A inhibitor approved by the US Food and Drug Administration (FDA) and European Medicines Agency for treatment of moderate-to-severe psoriasis and psoriatic arthritis (PsA) in adults [2-4]. In May 2019, it was approved and marketed in mainland China.
IL-17A plays a pivotal role in the pathogenesis of psoriasis. IL-17A is produced by T-helper (Th)17 cells and innate immune cells [5]. Studies have shown that Th17 cells and IL-17 may be involved in the pathogenesis of hepatitis caused by viral infection [6]. The hepatitis-B virus (HBV) is a hepadnavirus responsible for most liver diseases. Approximately 350–400 million people worldwide suffer from chronic infection with the HBV [7].
The safety of biologic therapy in patients with HBV infection is controversial. Biologic therapy in patients with psoriasis has been reported to be related to HBV reactivation [8]. There are few safety data for secukinumab in psoriasis patients with HBV infection because clinical trials typically provide evidence from patients without certain comorbidities or infections that may render them immuno-compromised.
We conducted a retrospective study to assess the safety and efficacy of secukinumab therapy in psoriasis patients with HBV infection in a real-life scenario.
Methods
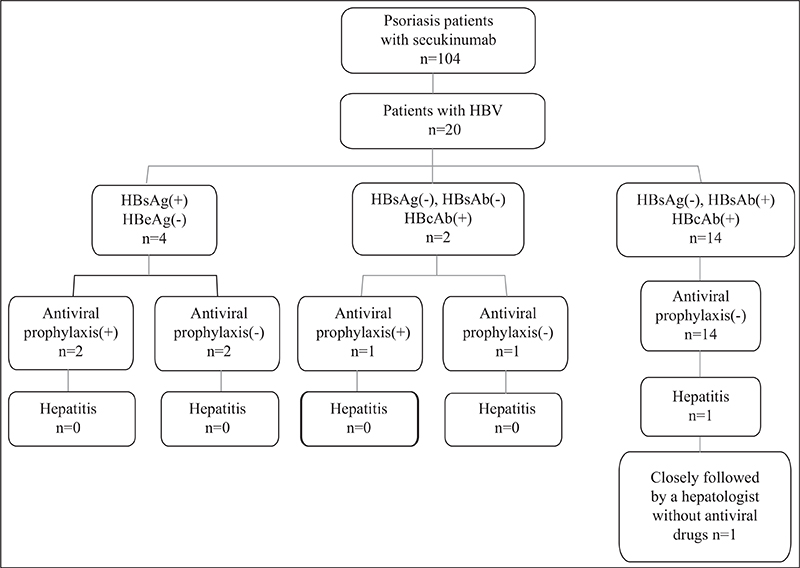
This retrospective study included all psoriasis patients with HBV infection treated with secukinumab from May 2019 to September 2020 at Shanghai Skin Disease Hospital (Shanghai, China). All patients had been treated with secukinumab for ≥24 weeks. Secukinumab (300 or 150 mg, s.c.) was administered at Weeks 0, 1, 2, 3 and 4, and every four weeks thereafter. The secukinumab dose was determined by a dermatologist on the basis of body weight and the financial situation of the patient. In China, 5–7.99% of the population are positive for the surface antigen of the hepatitis B virus (HBsAg) [9]. All patients treated with secukinumab should undergo pre-screening for HBV markers. The demographic and serological findings of patients, such as levels of HBV markers and HBV-DNA load, were detected at baseline and Week 24. The scores for the Psoriasis Area and Severity Index (PASI), percent of affected body surface area (BSA), Investigator Global Assessment (IGA), Dermatology Life Quality Index (DLQI), and alanine transaminase (ALT) level and aspartate transaminase (AST) level were monitored at Weeks 0, 4, 8, 12 and 24 during secukinumab treatment.
The National Psoriasis Foundation (NPF) has divided hepatitis B serologic status into six categories: (I) uninfected (HBsAg−, HBsAb−, HBcAb−, HBeAg− and HBeAb−); (II) vaccinated (HBsAg−, HBsAb+, HBcAb−, HBeAg −and HBeAb−); (III) acute HBV infection (HBsAg+, HBsAb−, HBcAb+, HBcIgM+); (IV) chronic HBV infection, which can be subdivided into chronic active HBV infection (HBsAg+, HBsAb−, HBcAb+, HBeAg+ and HBeAb−) and chronic inactive HBV infection (HBsAg+, HBsAb−, HBcAb+, HBeAg− and HBeAb+); (V) occult HBV infection (HBsAg−, HBsAb−, and HBcAb+); and (VI) resolved HBV infection (HBsAg−, HBsAb+, and HBcAb+) [10].
HBV virological reactivation was defined as an increase in the HBV-DNA load of at least 1 log 10 copies/mL or a switch from undetectable to detectable after secukinumab treatment [11]. “Hepatitis” was defined as a ≥three-fold increase in the ALT level that exceeded the upper limit of normal [12]. Patients with chronic active and inactive HBV infection are suggested to receive antiviral prophylaxis prior to immunosuppressive drug therapy [10]. However, based on the policy of the National Healthcare Security Administration (NHSA) in China, antiviral prophylaxis is restricted to patients with active HBV infection. The NHSA does not reimburse the cost of antiviral therapy for patients not suffering from active HBV infection who receive biologic agents. Hence, most patients without active HBV infection refused to accept antiviral prophylaxis. If patients declined prophylaxis, they were reviewed regularly by a hepatologist.
Ethics approval was obtained from the Ethics Committee of Shanghai Skin Disease Hospital (SSDH-IEC-SG-029-3.1).
Statistical analysis
The two-tailed Student’s t-test was used to compare the scores for the PASI, % BSA affected, IGA and DLQI between baseline and Week 4, or baseline and Week 24, respectively. Data were presented as mean ± SD, and p < 0.05 was considered significant. Statistical analysis was undertaken using Prism 7 (GraphPad, San Diego, CA, USA).
Results
Patients and characteristics
A total of 104 consecutive patients with psoriasis receiving secukinumab therapy were screened. Finally, the data for 20 patients (14 males and six females) with HBV infection and 24 weeks of secukinumab treatment were included. Data on demographics and treatment are summarized in table 1. These 20 patients had a mean duration of psoriasis of 18.65 years, 15% had a family history of psoriasis, 50% were smokers/consumed alcohol, 10% were obese, and one patient had PsA. One or more comorbidities were present in 30% patients, the most common being diabetes mellitus (20%) and hypertension (15%), and one patient had fatty liver. Notably, 90% of patients were naïve to biologic therapy. Ninety percent of patients had conventional treatment, with traditional Chinese medicine (TCM) formulations being the most common (60%), followed by acitretin (25%), phototherapy (20%) and methotrexate (15%).
HBV infection
Four of the 20 patients (20%) had chronic inactive HBV infection; the HBV-DNA load was
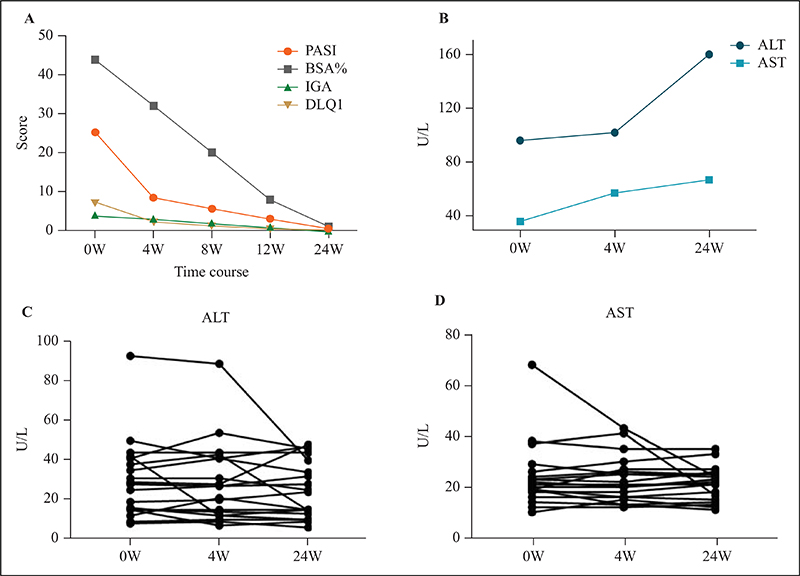
The serology findings of all HBV-infected patients are summarized in figure 1 and table 2. Among all patients who received secukinumab, 19 patients showed a steady and normal level of liver enzymes after 24 weeks of therapy (figure 2C, D). However, one patient with resolved HBV infection and fatty liver with elevated baseline liver enzymes experienced hepatitis. The clinical course of this patient is shown in figure 2A, B. Briefly, during 24 weeks of treatment, the PASI score (25.2, 8.4, 5.6, 3.0 and 0.4 at Weeks 0, 4, 8, 12 and 24, respectively), % BSA affected (44, 32, 20, 8 and 1 at Weeks 0, 4, 8, 12 and 24, respectively), IGA score (4, 3, 2, 1 and 0 at Weeks 0, 4, 8, 12, and 24, respectively) and DLQI score (7, 2, 1, 0 and 0 at Weeks 0, 4, 8, 12, and 24, respectively) of the patient decreased significantly, whereas the levels of ALT (U/L) (96, 102 and 160, at Week 0, 4, and 24, respectively) and AST (U/L) (36, 57 and 67 at Weeks 0, 4 and 24, respectively) increased after 24 weeks of secukinumab treatment. Notably, before secukinumab therapy, this patient had baseline levels of liver enzymes that were slightly above the upper limit of normal. He had a history of fatty liver and the HBV-DNA load was
Efficacy of secukinumab treatment in patients with psoriasis and HBV infection
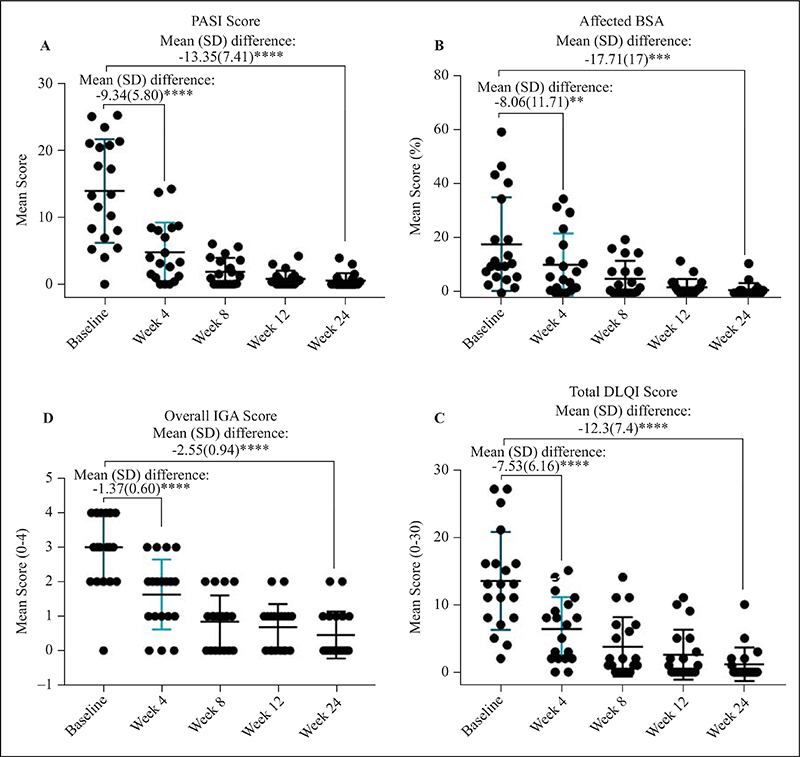
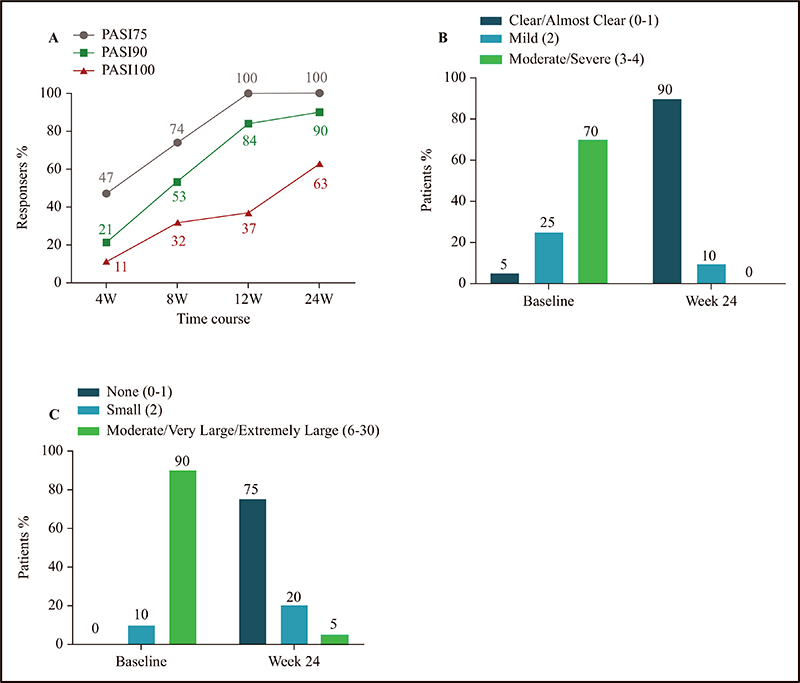
Secukinumab treatment improved the scores of PASI, DLQI and IGA, and % BSA affected significantly from baseline to 24 weeks of treatment, and showed dramatic changes at Week 4 (figure 3). From Week 0 to Week 24, patients showed a significant improvement in the PASI score (−13.35 ± 7.41, p < 0.0001), % BSA affected (−17.11 ± 17, p = 0.0002), IGA score (−2.55 ± 0.94, p < 0.0001), and DLQI score (−12.3 ± 7.39, p < 0.0001). From Week 0 to Week 4, patients showed a significant improvement in the PASI score (−9.34 ± 5.80, p < 0.0001), % BSA affected (−8.06 ± 11.71, p = 0.0077), IGA score (−1.37 ± 0.60, p < 0.0001), and DLQI score (−7.53 ± 6.16, p < 0.0001). The proportion of patients with PASI 75 (47%, 74%, 100%, 100% and 0 at Weeks 0, 4, 8, 12, and 24, respectively), PASI 90 (21%, 53%, 84%, 90% and 0 at Weeks 0, 4, 8, 12, and 24, respectively) and PASI 100 (11%, 32%, 37%, 63% and 0 at Weeks 0, 4, 8, 12, and 24, respectively) are shown in figure 4A. The proportion of patients with clear/almost clear status (IGA score = 0–1) was significantly higher at 24 weeks of treatment than at baseline (5% and 90% at Week 0 and Week 24, respectively). The proportion of patients with moderate/severe status (IGA score = 3–4) was significantly lower at 24 weeks of treatment than at baseline (70% and 0% at Week 0 and Week 24, respectively) (figure 4B). The proportion of patients who reported no impact of disease on quality of life (DLQI score = 0–1) was significantly higher at 24 weeks of treatment than at baseline (0% and 75% at Week 0 and Week 24, respectively). Accordingly, the proportion of patients who reported a moderate-to-extremely-large impact of disease on quality of life (DLQI score = 6–30) was significantly lower (90% and 5% at Week 0 and Week 24, respectively) (figure 4C).
Discussion
We show that secukinumab was highly efficacious in psoriasis patients with HBV infection. In accordance with observations from clinical trials [13, 14], secukinumab showed rapid action in our study, with significant improvements in psoriasis disease and indices of quality of life reported after four weeks of treatment.
The approval of secukinumab heralded a new era in psoriasis management. It shifted possible outcomes of psoriasis treatment from 75% reduction of the PASI score (PASI75) to PAS I90, and even complete resolution of skin lesions (PASI 100), which are now recognized goals for psoriasis improvement [15, 16]. Numerous factors should be taken into account when selecting appropriate therapy for a psoriasis patient with HBV infection, including commonly associated comorbidities (e.g. obesity, psoriasis arthritis and cardiovascular disease), patient age and concomitant infections [17, 18]. Most of our patients had experience of using TCM formulations but not biologic agents. Hence, cost and reimbursement were important factors when choosing clinical treatment.
Levels of the liver enzymes, ALT and AST, of most patients were stable over 24 weeks of treatment with secukinumab, in contrast to etanercept use, which increased the levels of these enzymes after Week 16; data which are consistent with results from studies using anti-tumour necrosis factor-α agents [19]. One patient with resolved HBV infection experienced hepatitis. This patient had levels of liver enzymes slightly above the upper limit of normal before secukinumab therapy, and a five-year history of fatty liver. The hepatologist suggested more exercise and diet control, and after six months of change in life-style, his BMI and abdominal girth decreased, and his liver enzymes returned to normal value without antiviral drugs. During these six months, he continued secukinumab management. Thus, poor baseline liver function, without any intervention, might influence side effects of secukinumab, and liver enzymes and HBV load should be monitored closely in these patients.
Biologic agents can disturb the delicate balance between the degree of virus replication and immune control of the host, which can lead to virus reactivation. Emerging data have shown that IL-17 is involved in the pathogenesis of HBV infection and liver injury by inducing pro-inflammatory and pro-fibrotic cytokines [20]. Few studies have evaluated the safety of secukinumab for treatment of psoriasis patients with HBV infection [11, 21, 22]. A recent review by Snast and colleagues [11] focused on 40 patients with chronic HBV infection treated with different biologic therapies. They claimed that biologic therapies were a considerable risk in patients with chronic HBV infection; eight patients (20%) developed viral activation and one patient had concurrent hepatitis.
The largest study to assess the safety of secukinumab treatment in psoriasis patients with concomitant HBV infection (n = 49) [21] revealed that six patients (27%) with chronic HBV infection who did not undergo antiviral treatment experienced viral reactivation. In addition, no patients with chronic HBV infection who had antiviral therapy experienced viral reactivation. One patient (9%) with occult HBV infection who did not undergo antiviral treatment experienced viral reactivation. The authors concluded that patients with chronic and occult HBV infection can develop viral reactivation during secukinumab therapy. In our study, none of the patients with or without antiviral treatment showed virus reactivation. This might be due to the smaller sample size of our study; large-scale studies are needed to validate our conclusions. Besides, the interval between viral load monitoring, the duration and dose of biologic therapy and concurrent use of immunosuppressants can account for the differences in the incidence of reactivation. According to NFP recommendations, in uninfected or vaccinated individuals, the use of immunosuppressive drug therapy (ISDT) is permitted. For acute HBV infection, ISDT should be deferred. For patients with chronic active and inactive HBV infection, antiviral prophylaxis before ISDT is highly recommended. For occult and resolved HBV infection, antiviral prophylaxis should be administered in consultation with a hepatologist in case of HBV reactivation [10]. Whereas, for patients receiving secukinumab, current recommendations for managing HBV infection are still based on previously approved biologics, such as anti-TNF-α agents. In previous studies including our own, not all patients with chronic inactive HBV infection received antiviral therapy during secukinumab treatment. This scenario underscores the need to unify criteria for the indication of antiviral prophylaxis in psoriasis patients undergoing treatment with new biologic therapies, and reflects that further studies should be conducted to demonstrate the safety of relatively new biologics.
This study’s limitations include the small sample size as well as the lack of a comparison group, such as psoriasis patients with HBV infection treated without immunosuppressive drugs.
Conclusions
Secukinumab showed good efficacy in psoriasis patients with HBV infection. Chronic inactive, occult and resolved HBV infection might not increase the risk of hepatitis during secukinumab treatment. However, patients with poor baseline liver function, without any intervention during secukinumab treatment, might experience hepatitis. Periodic monitoring including HBV markers, HBV-DNA load and serological liver-function tests are necessary during secukinumab treatment.
Acknowledgments and disclosures
Acknowledgments: The patients in this manuscript have given written informed consent to publication of their case details.
Funding: This work was sponsored by grants from National Natural Science Foundation of China (No. 81872522, 82073429, 82003334), Innovation Program of Shanghai Municipal Education Commission (No.2019-01-07-00-07-E00046), the Program of Science and Technology Commission of Shanghai Municipality (No. 18140901800), Excellent Subject Leader Program of Shanghai Municipal Commission of Health and Family Planning (No. 2018BR30), Clinical Research Program of Shanghai Hospital Development Center (No. SHDC2020CR1014B, SHDC12018X06) and Program of Shanghai Academic Research Leader (No. 20XD1403300), Shanghai Health and Family Planning Commission (201840365).
Conflicts of interest: none.