Hématologie
MENUOptimising the evaluation of therapeutic response to first-line treatment of diffuse large B cell lymphoma Article à paraître
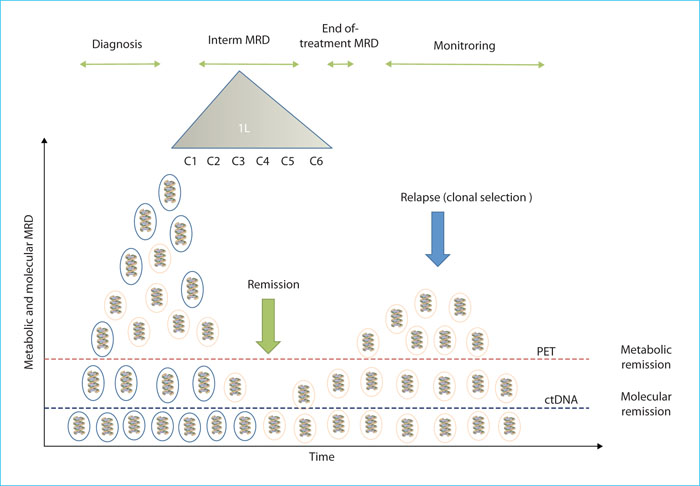
The concept of minimal residual disease (MRD) has taken centre stage in haematology in recent years. Due to technological advances, interest in it is no longer limited to the historical field of acute leukaemia but has extended to all haematological malignancies such as myeloma, chronic lymphocytic leukaemia and chronic myeloproliferative syndromes. Lymphomas and especially diffuse large B-cell lymphomas (DLBCL) are no exception to this, and within this context, there are two objectives: on the one hand, to best evaluate the kinetics and quality of the response during or at the end of treatment, and on the other, to allow the treatment to be adjusted according to MRD. In order to achieve this, clinicians have two major tools at their disposal. One forms part of their routine activity: metabolic imaging using positron emission tomography (PET), at either an interim stage (iPET) or at the end of the treatment. The other, which is still emerging, is based on the quantification of circulating plasma tumour DNA (ctDNA). “Metabolic” MRD and “molecular” MRD are, therefore, the two approaches currently used to assess the therapeutic response of patients with DLBCL (figure 1). In this review, we look at the strengths and limitations of these approaches, as well as their complementarity in the first-line management of DLBCL in 2021.
The use of positron emission tomography in assessing therapeutic response
The 2014 Lugano criteria are the most recent recommendations for the initial grading and assessment of response to treatment and monitoring of lymphoma, replacing the outdated criteria formulated by the International Working Group (IWG) in 2007 [1]. It should be recalled that PET/CT is the most effective method to identify and monitor ganglion and extra-ganglion node sites, with the exception of central nervous system involvement, for which MRI remains the reference.
Improving and standardising response criteria
PET during early assessment is also an important step in the management of DLBCL, although there is no consensus around recommendations outside clinical trials. The original work assessing the value of iPET was based on qualitative visual criteria (positive/negative), leading to a lack of reproducibility and poorly reproducible results. The validity of iPET+ has been brought into question by systematic biopsies, leading to the identification of lymphomatous disease in only 23% of cases [2]. This low predictive value has led to the use of quantitative or semi-quantitative methods to limit the risk of false positives.
Assessing the difference in maximum standardised uptake value (ΔSUVmax) between two examinations and the Deauville criteria have thus allowed for a very significant improvement in the specificity and predictive values of iPET [3-5].
Measuring ΔSUVmax requires imaging on identically calibrated machines, a SUVmax basal state of at least 10, and a residual SUVmax >5 (Menton criteria, 2011). It is a reproducible method and has already been applied in the PETAL and GAINED multi-centre clinical trials.
Current recommendations are to use the Deauville Scale (DS), established in 2009, which allows five-point grading of residual lesions, regardless of their size (table 1). The DS can be used for interim and end-of-treatment evaluations. A score of 1, 2 or 3 is considered to be a complete metabolic response, even if there is residual mass.
In order to improve the DS, some authors have suggested the use of a quantitative method combined with the DS, in particular to better distinguish between DS 3 and DS 4. The Ration Deauville Scale (rDS) was thus defined, based on the measurement of the ratio (r) of SUV between the tumour and the liver (right lobe). The advantage of the rDS is that it is independent of injected activity and weight, allowing a shift from a discontinuous semi-quantitative scale (DS) to a continuous semi-quantitative scale, combining the positive aspects of DS and ΔSUVmax. In a single-centre cohort of 181 first-line DLBCL patients, an rDS >1.4 (i.e., residual mass with SUVmax >141% of liver SUVmax) after four cycles (C4) and at the end of treatment appeared to outperform DS in terms of positive predictive value, prediction of progression-free survival (PFS) and prediction of overall survival (OS). Furthermore, in multivariate analysis, only rDS retained a predictive value after C4 or at the end of treatment, compared to SD or ΔSUVmax[6].
Although other studies have suggested different ratios after C2 (1.25) or C4 (1.6) [7, 8], this approach is clearly an interesting way to identify the most severe patients, especially when combined with the international prognostic index (IPI) [6].
Optimal time to perform iPET
The majority of data in the literature concerning iPET are derived from post-C2 or -C4 assessments, more rarely post-C1 or -C3 [9]. Based on a meta-analysis of 1,977 DLBCL cases included in different prospective trials, PET after C2 seems to be most effective in identifying the best responders. In contrast, post-C4 PET predicts worst-case PFS and is the most accurate prognosticator, suggesting that the ideal time to assess the patient and adjust the treatment strategy [10] is after the fourth treatment. Depending on the response criteria used, the negative predictive value for PFS after two or four cycles ranges from 67% to 100%, while the positive predictive value ranges from 36% to 100% [9].
Adjusting treatment according to iPET
The clinical relevance of early assessment (post-C2 or -C4) is illustrated by the possibility of de-escalating therapy, as in Hodgkin's lymphoma.
The LNH2007-3B trial, conducted under the auspices of the Lymphoma Study Association (Lysa), showed that an intensification of treatment with peripheral stem cell autotransplantation could be avoided in up to 25% of cases for DLBCL with an age-adjusted IPI (aaIPI) of 2–3. Indeed, the prognosis of patients with an ΔSUVmax >66% and >70% after C2 and C4, respectively, was comparable to that of chemosensitive patients consolidated by autograft [11].
In the Goelams LNH-02-03 Phase III trial, dedicated to non-bulky localised forms, radiotherapy could be omitted without compromising the prognosis for PET-negative patients after four cycles of rituximab, cyclophosphamide, doxorubicin, vincristine and prednisone (R-CHOP) [12]. This clinical trial data is supported by studies from our Canadian colleagues who recently published their observations based on a retrospective study during routine practice to assess the value of performing “consolidating” radiotherapy in the case of a positive end-of-treatment PET after six R-CHOPs. This was an uncontrolled study with many biases, but involved 723 patients with a median follow-up of 4.3 years. The authors also concluded that “consolidating” radiotherapy may not be performed if the end-of-treatment PET is negative, which also applies to patients who have “bulky” disease (>10 cm) [13]. The results of the Lysa LNH-091B trial, which compares six to four R-CHOPs in patients with an IPI = 0, are awaited and should shed light upon the value of PET-guided de-escalation.
While “metabolic MRD” provides arguments for de-escalation, it remains to be seen whether it can, a contrario, indicate a strategy for therapeutic intensification. The Phase III PETAL trial compared a standard arm (R-CHOP14 × 6) to an experimental arm based on Burkitt-like escalation if the post-C2 iPET showed an ΔSUVmax <66%. In this trial, 13% of patients were iPET positive and showed a much worse prognosis. However, despite the intensification of therapy, these patients did not satisfactorily recover using the proposed strategy [14, 15].
Circulating plasma tumour DNA and molecular minimal residual disease
While the measurement of ctDNA is an undeniable step forward in assessing MRD in DLBCL, its limitations and drawbacks should be noted [16].
Pre-analytical constraints
Several methods are currently available, but in all cases, these require rigorous pre-analytical procedures. Circulating plasma DNA is fragile and degrades rapidly. Thus, it is recommended not to exceed three to six hours between collection, centrifugation and storage of aliquots (at -80 °C), even if some tubes (e.g. Streck tubes) allow for longer conservation and are suitable for delayed dispatch for centralised analysis. Although the correlation between ctDNA and tumour mass is well established, particularly in DLBCL, certain factors are likely to influence the concentration of ctDNA independently of it, such as inflammatory status, physical effort or circadian variation. Plasma is preferred to serum, which is richer in non-tumour DNA [17].
Available technologies
The most commonly used methods include sequencing of variable regions of VDJ immunoglobulin genes (clonotypic sequences of heavy and/or light chains of immunoglobulin genes) (adaptive), sequencing after gene identification and/or amplification using a targeted gene panel (AmpliSeq, QiaSeq), and digital PCR (dPCR) adapted to a limited number of targeted known recurrent mutations (e.g. the MYD88L265P variant) [18-20]. Among the most promising approaches used in the context of DLBCL, personalised profiling of cancer by deep sequencing (CAPP-Seq) should also be mentioned. To date, this is the most sensitive and specific technique, allowing the identification of VDJ regions, mutants and recurrent translocations. CAPP-Seq provides very accurate ctDNA profiling and can predict the cell of origin (COO) (GCB/ABC) profile with 80% agreement with the Hans model and 86% agreement with fluorescent in situ hybridisation (FISH) data [21]. The low amount of ctDNA, the background of non-tumour circulating DNA and the technical artefacts of PCR make the interpretation of next-generation sequencing (NGS) data challenging and the sensitivity levels variable between techniques. The now widespread use of molecular markers, known as unique molecular identifiers (UMIs), has significantly improved the sensitivity of NGS in this context.
Molecular response measured by ctDNA quantification
The dynamics of ctDNA decay under treatment is a major prognostic factor. Patients who are negative after two cycles have a significantly higher PFS than those who are still positive (80.2 versus 41.7%; p <0.0001) [20]. These initial data may be refined by distinguishing between patients with major molecular response (MMR)–corresponding to a 2.5-log reduction in ctDNA after two cycles (C2)–and those with early molecular response (EMR)–defined by a 2-log reduction after C1. Importantly, MMR and EMR are independent prognostic factors for IPI and metabolic response, suggesting that ctDNA is indeed complementary to metabolic MRD [22]. These data predict PFS and OS after first-line therapy and suggest the implementation of an adaptive treatment strategy [22].
Combined and dynamic approaches
Kurtz et al. proposed an attractive multiparametric and dynamic approach to predicting prognosis by combining four factors into a specific index:
- –IPI
- –PET
- –molecular subtype (COO, GCB/ABC)
- –ctDNA in the form of three parameters:
- –baseline ctDNA concentration
- –EMR status
- –MMR status
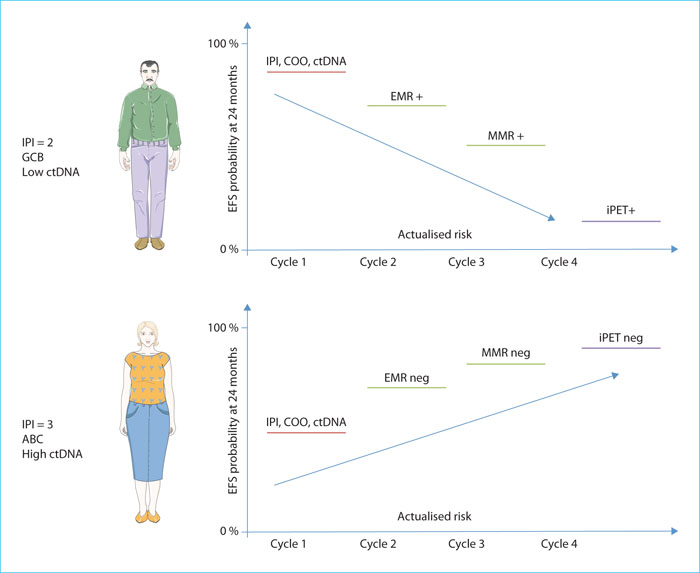
This index, known as the continuous individualised risk index (CIRI), is based on an initial probability of PFS at 24 months obtained before any treatment and is adapted by updating additional information collected during treatment. This probabilistic model provides a better prediction of PFS than any of its components, with notably fewer patients classified as high risk compared to identification based solely on IPI, COO or PET. Figure 2 illustrates two clinical situations where the probability of PFS at 24 months changes over the course of treatment and according to the information collected. The CIRI remains robust even with missing data. This approach, although relatively complex, is a novel attempt to integrate the key elements of patient prognosis [23]. It is likely that this model will be revisited in the light of what is now known about the baseline metabolic volume and, in particular, its complementarity with the biological characteristics of the tumour [24, 25].
Conclusion and outlook
In the era of targeted therapies and with the emergence of chimeric antigen receptor T cells (CAR-T) being included earlier in treatment lines, optimising the evaluation of therapeutic response is a major challenge in the management of DLBCL. The measurement of ctDNA and metabolic response are the two keys to this evaluation. These non-invasive techniques can be combined and repeated at key points in the therapeutic phases, mainly post-C2 and -C4 during the first line of treatment. An adaptive prognostic model could be proposed, provided that the response criteria are standardised and its relevance is demonstrated in Phase III trials.
Conflicts of interest
The author declares no conflicts of interest.