European Journal of Dermatology
MENUSurveillance of dermo-cosmetic products: a global cosmetovigilance system to optimise product development and consumer safety Volume 31, numéro 4, July-August 2021
- Mots-clés : dermato-cosmetics, undesirable effect, signal detection, patch test, allergy, adverse skin reaction
- DOI : 10.1684/ejd.2021.4101
- Page(s) : 463-9
- Année de parution : 2021
Background
In the absence of formal marketing authorisation, the manufacturers of cosmetic products are responsible for their compliance with the cosmetic regulations.
Objectives
To present the key features of a structured, reactive, and rigorous global cosmetovigilance system through practical examples.
Materials & Methods
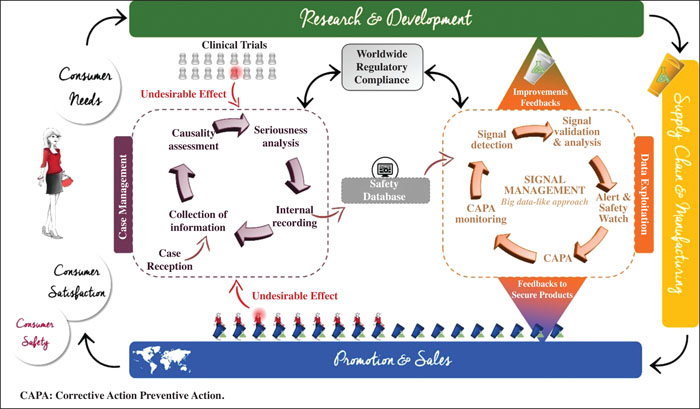
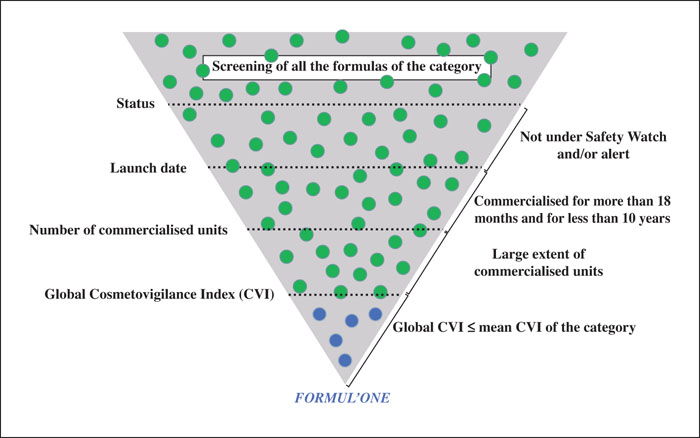
During clinical development, adverse reactions are collected formally and analysed by cosmetovigilance experts. After commercialisation, information on reported adverse reactions is sought directly from the consumers. The results of allergological investigations are systematically requested. Pre- and post-marketing cases are analysed along with other sources of information (e.g. monitoring of the literature) to detect safety signals per product and per ingredient. A cosmetovigilance index (CVI) is calculated for each formula, based on the number of cases, causality level and number of commercialised units. Updated periodically, it is used to detect signals and select the best tolerated formulas to help formulating new products.
Results
Examples of safety issues raised during development or after commercialisation, and corresponding corrective actions, are presented. These actions include (but are not limited to) a safety watch to closely monitor adverse reactions, the modification of the formula or a change in the packaging. Cosmetovigilance data also impact future product development, as illustrated by the work done on sunscreens.
Conclusion
Through the rigorous collection and analysis of adverse reactions during development and after commercialisation, the safety of dermo-cosmetic products can be improved by taking the appropriate corrective actions, monitoring their effectiveness and optimising future product development by focusing on the best tolerated formulas.