European Journal of Dermatology
MENUSafety and efficacy of a carboxymethyl chitosan dermal injection device for the treatment of skin defects: a first-in-man, pilot, comparative, split-body study Volume 31, numéro 4, July-August 2021
- Mots-clés : ageing skin, carboxymethyl chitosan, injectable soft-tissue device, non-animal origin
- DOI : 10.1684/ejd.2021.4091
- Page(s) : 549-58
- Année de parution : 2021
Background
Injectable soft-tissue devices are increasingly used for improving skin defects and deficiencies related to ageing.
Objectives
To assess the safety and efficacy of KIO015, a new injectable soft-tissue device formulated with carboxymethyl chitosan for the intradermal treatment of skin defects associated with ageing.
Materials & Methods
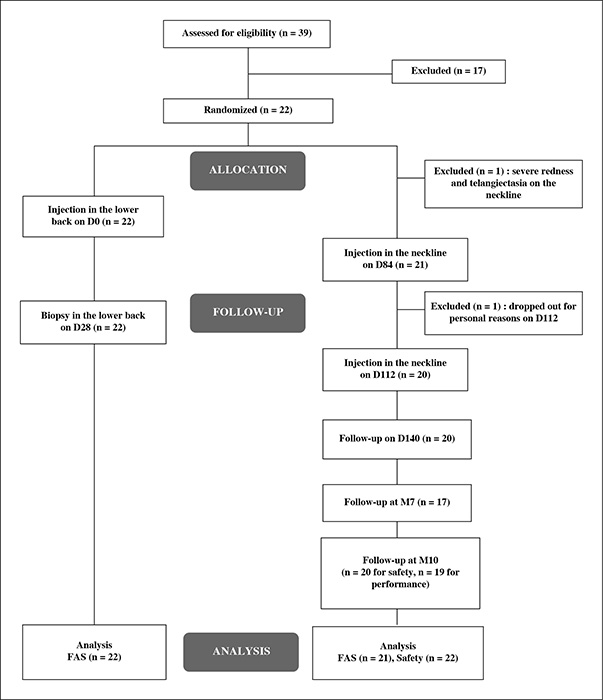
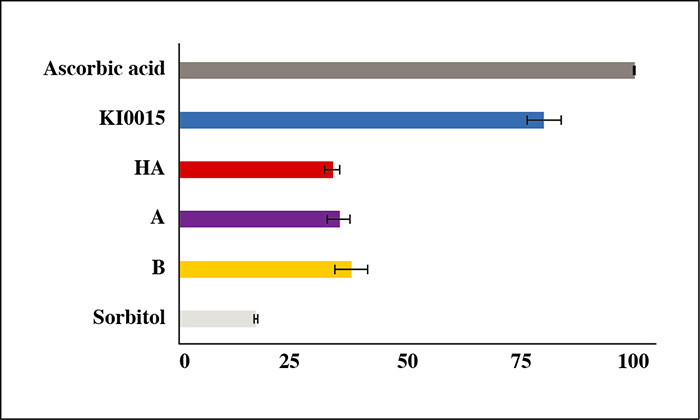
Twenty-two subjects (40-65 years) were randomized to receive injections in the neckline of KIO015 and a non-cross-linked HA-based device, and were followed for up to 10 months. Injection site reactions (ISRs) and adverse events (AEs) were documented. Skin improvement was assessed instrumentally and clinically. Skin biopsies at injection zones in the lower back were taken at Day 28 for histopathology and immunohistochemistry analyses, to further assess product performance. Histomorphometric analyses on rabbits and in vitro assessment of KIO015 antioxidant capacity were also conducted.
Results
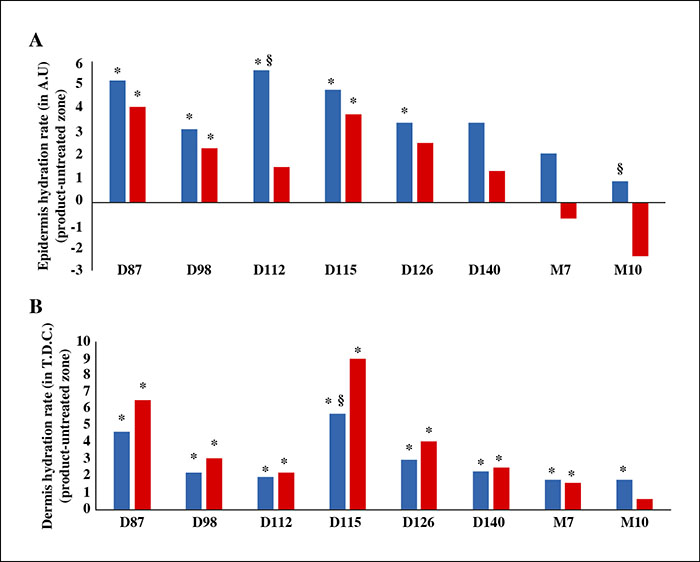
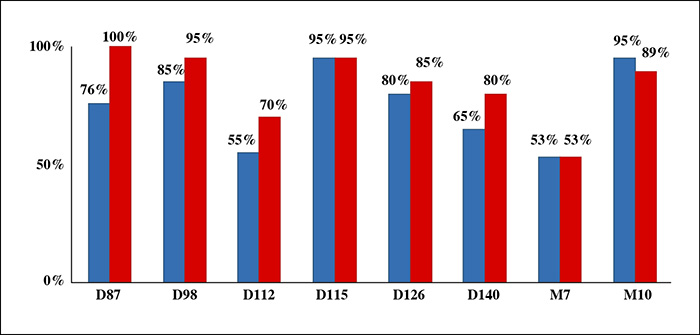
KIO015 was very well tolerated. Only expected and transient ISRs were observed; mainly erythema and hematoma. No adverse local effects or foreign body granuloma were observed histologically. Both clinical and instrumental evaluations confirmed the performance of KIO015. The skin was firmer and more elastic. Skin hydration showed significant improvement three days after injection. KIO015 exhibited superior overall maintenance of skin hydration after 10 months as compared to HA. These clinical results were supported by in vitro trials and implantation tests in the rabbit.
Conclusion
The results from this pilot study support the use of KIO015 as an innovative alternative to HA-based devices for intradermal treatment of skin disorders