European Journal of Dermatology
MENUReal-world data on the use of secukinumab as treatment for moderate-to-severe psoriasis in Chinese patients Volume 30, numéro 5, September-October 2020
- Mots-clés : psoriasis, secukinumab, efficacy, safety
- DOI : 10.1684/ejd.2020.3878
- Page(s) : 554-60
- Année de parution : 2020
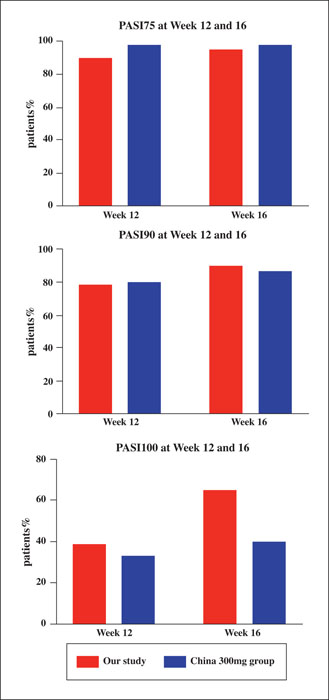
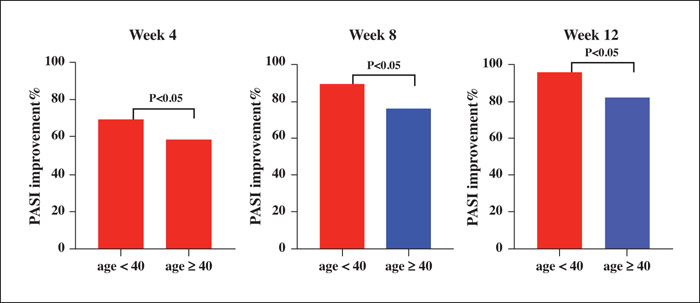
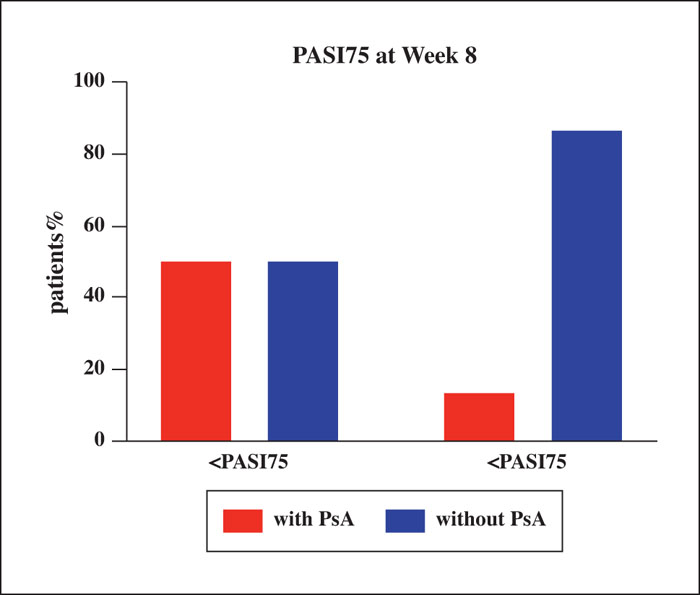
Background: The efficacy and safety of secukinumab, an interleukin-17 inhibitor, as systemic treatment for patients with moderate-to-severe psoriasis have been demonstrated, but real-world data pertaining to this is limited in China. Objective: To evaluate the efficacy and safety of secukinumab in clinical practice in Chinese psoriasis patients with or without psoriatic arthritis (PsA) and identify potential baseline factors that affect the response of patients to secukinumab treatment. Materials & Methods: Data from 81 patients treated with secukinumab for at least 16 weeks were analysed in a retrospective observational study. Results: After 16 weeks of treatment with secukinumab, 91.1%, 73%, and 38.3% of patients achieved a PASI 75 (75% improvement based on the Psoriasis Area and Severity Index), PASI 90, and PASI 100, respectively. A significant improvement in the quality of life of patients was also observed. Notably, baseline factors, such as young age, lower BMI, no scalp involvement and absence of concomitant PsA, were associated with better clinical response to secukinumab. Approximately 42% of patients (34/81) experienced adverse events, of which the most common was pruritus. Conclusion: The results demonstrated that secukinumab appears to be an effective treatment alternative for the majority of Chinese plaque psoriasis patients. Baseline factors, including age, BMI, scalp involvement and concomitant presence of PsA, were associated with response to secukinumab.