European Journal of Dermatology
MENUA new case series on etanercept treatment for toxic epidermal necrolysis Volume 30, numéro 5, September-October 2020
- Mots-clés : etanercept, therapy, toxic epidermal necrolysis (TEN)
- DOI : 10.1684/ejd.2020.3883
- Page(s) : 561-8
- Année de parution : 2020
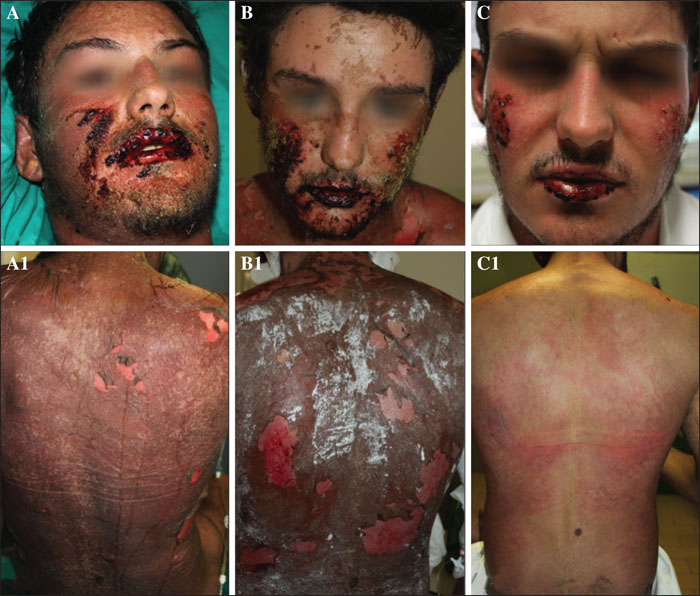
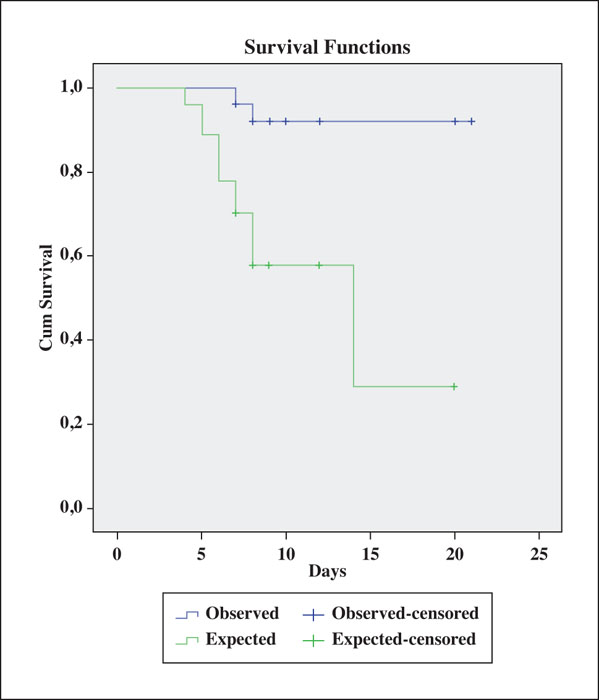
Background: Toxic epidermal necrolysis (TEN) is a severe, potentially lethal drug reaction for which no standard treatment is available. Objective: To describe 17 consecutive TEN patients treated with a single dose of etanercept, a TNF-alpha inhibitor. Materials & Methods: Comorbidities and any drug treatment initiated within the previous month were recorded on admission. Patients received 50 mg etanercept in a single subcutaneous injection. The clinical severity of the disease was computed using the SCORTEN scale. The expected number of deaths was calculated based on the probability of death associated with each SCORTEN score level. Healing was defined as complete re-epithelialization. Time to healing was analysed using the Kaplan-Meier estimator. Results: The lowest SCORTEN score was 2, and seven patients scored in the most severe risk category (i.e., =>5). A comparison between observed (2/17) vs. expected deaths (10/17) was statistically significant (p=0.012). Fifteen patients promptly responded to treatment and achieved complete re-epithelization (median time to healing: 8.5 days), without complications or side effects. The two observed deaths were due to other causes, although re-epithelization had initiated in both patients. Conclusion: These preliminary results add to our initial observations indicating that etanercept may effectively control TEN, a potentially lethal skin condition for which there is currently no effective cure. Where funding is available, randomized controlled trials on etanercept for TEN should be conducted.