Epileptic Disorders
MENUDiagnostic value of MRI in the presurgical evaluation of patients with epilepsy: influence of field strength and sequence selection: a systematic review and meta-analysis from the E-PILEPSY Consortium Volume 24, numéro 2, April 2022
- Mots-clés : magnetic resonance imaging, lesion, diagnostic imaging, refractory epilepsy
- DOI : 10.1684/epd.2021.1399
- Page(s) : 323-42
- Année de parution : 2022
Objective
MRI is a cornerstone in presurgical evaluation of epilepsy. Despite guidelines, clinical practice varies. In light of the E-PILEPSY pilot reference network, we conducted a systematic review and meta-analysis on the diagnostic value of MRI in the presurgical evaluation of epilepsy patients.
Methods
We included original research articles on diagnostic value of higher MRI field strength and guideline-recommended and additional MRI sequences in detecting an epileptogenic lesion in adult or paediatric epilepsy surgery candidates. Lesion detection rate was used as a metric in meta-analysis.
Results
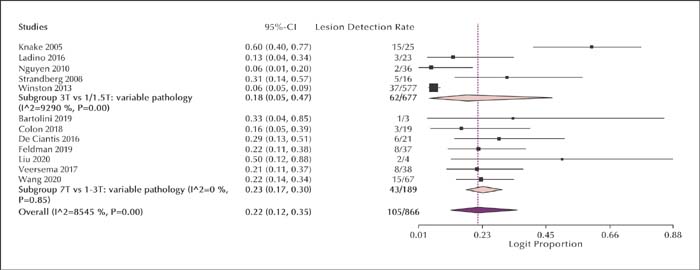
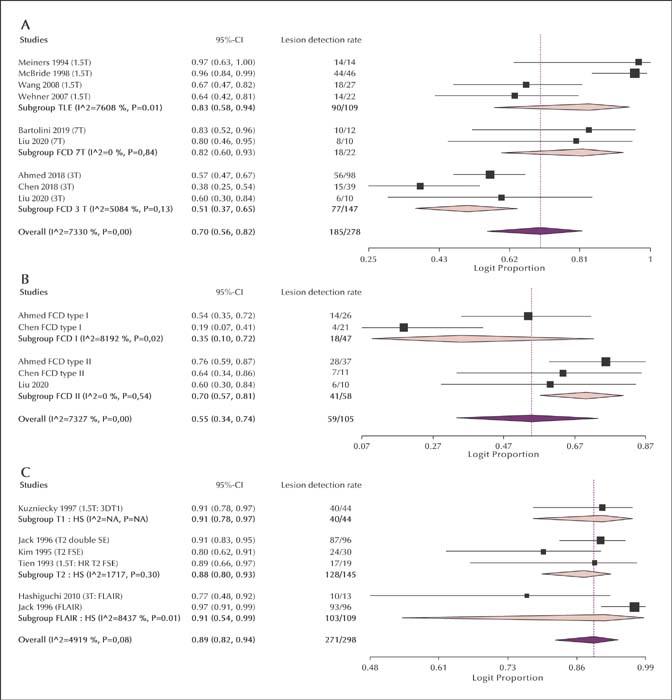
Eighteen studies were included for MRI field strength and 25 for MRI sequences, none were free from bias. In patients with normal MRI at lower-field strength, 3T improved lesion detection rate by 18% and 7T by 23%. Field strengths higher than 1.5T did not have higher lesion detection rates in patients with hippocampal sclerosis (HS). The lesion detection rate of epilepsy-specific MRI protocols was 83% for temporal lobe epilepsy (TLE) patients. Dedicated MRI protocols and evaluation by an experienced epilepsy neuroradiologist increased lesion detection. For HS, 3DT1, T2, and FLAIR each had a lesion detection rate at around 90%. Apparent diffusion coefficient indices had a lateralizing value of 33% for TLE. DTI fractional anisotropy and mean diffusivity had a localizing value of 8% and 34%.
Significance
A dedicated MRI protocol and expert evaluation benefits lesion detection rate in epilepsy surgery candidates. If patients remain MRI negative, imaging at higher-field strength may reveal lesions. In HS, apparent diffusion coefficient indices may aid lateralization and localization more than increasing field strength. DTI can add further diagnostic information. For other additional sequences, the quality and number of studies is insufficient to draw solid conclusions. Our findings may be used as evidence base for developing new high-quality MRI studies and clinical guidelines.