Epileptic Disorders
MENUBreastfeeding while on treatment with antiseizure medications: a systematic review from the ILAE Women Task Force Volume 24, numéro 6, December 2022
- Mots-clés : epilepsy, breastfeeding, antiseizure medications, lactation
- DOI : 10.1684/epd.2022.1492
- Page(s) : 1020-32
- Année de parution : 2022
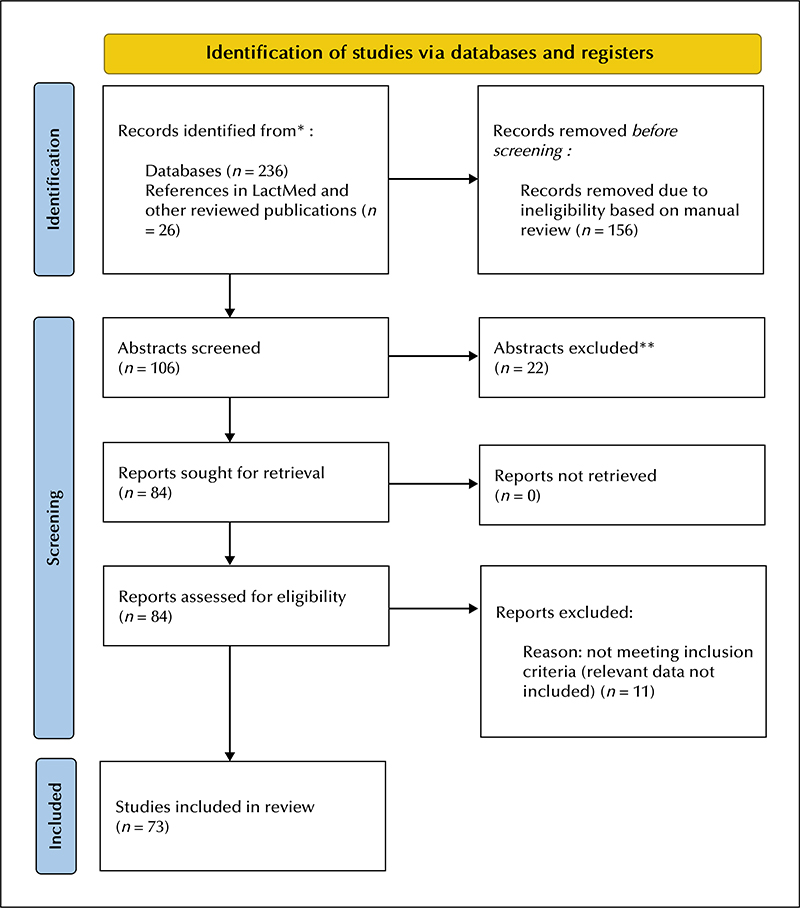
We carried out a systematic review of published information on transfer of antiseizure medications (ASMs) into breastmilk, ASM serum concentrations in breastfed infants, and the wellbeing of infants breastfed by mothers on ASM treatment. Information was extracted from 85 relevant articles. No data on ASM levels in breastmilk or in breastfed infants was identified for cannabidiol, cenobamate, clobazam, eslicarbazepine-acetate, everolimus, felbamate, fenfluramine, retigabine, rufinamide, stiripentol, tiagabine, and vigabatrin. For ASMs, with available information on levels in breastfed infants, very low concentrations (in the order of 10% or less of maternal serum concentrations) were reported for carbamazepine, gabapentin, levetiracetam, oxcarbazepine, phenytoin, valproate, and clonazepam. Slightly higher levels (up to approximately 30% of maternal serum concentrations) have been observed with lamotrigine and topiramate, and in single case reports for brivaracetam, lacosamide, and perampanel. High infant levels (30% up to 100% of maternal serum concentrations) have been reported with ethosuximide, phenobarbital and zonisamide. Adverse infant effects during breastfeeding by mothers on ASMs appear to be rare regardless of the type of ASM, but systematic study is limited. Prospective long-term follow-up studies of developmental outcomes among children who have been breastfed by mothers taking ASMs are sparse and have mainly involved children whose mothers were taking carbamazepine, lamotrigine, levetiracetam, phenytoin or valproate as monotherapy while breastfeeding. Although these studies have not indicated poorer outcome among breastfed children compared with those who were not breastfed, further data on long-term outcomes are needed to draw firm conclusions. It is concluded that breastfeeding should in general be encouraged in women taking ASMs, given the well-established benefits of breastfeeding with regard to both short- and long-term infant health in the general population. Counselling needs to be individualized including information on the current knowledge regarding the woman’s specific ASM treatment.
![]() Cette œuvre est mise à disposition selon les termes de la
Licence Creative Commons Attribution - ShareAlike 3.0 International
Cette œuvre est mise à disposition selon les termes de la
Licence Creative Commons Attribution - ShareAlike 3.0 International