European Journal of Dermatology
MENUExpert opinion: defining response to omalizumab in patients with chronic spontaneous urticaria Volume 27, numéro 5, September-October 2017
Grenoble, France
University Hospital and Faculty of Medicine,
University of Coimbra, Coimbra, Portugal
University Medical Center Utrecht,
Utrecht, The Netherlands
UZ Gent, Gent, Belgium
Hyderabad, India
Women's College Hospital, Toronto,
Canada; University of Toronto, Toronto, Canada
Bispebjerg Hospital & Department of Biomedical Sciences,
University of Copenhagen, Copenhagen, Denmark
- Mots-clés : chronic spontaneous urticaria, complete response, partial response, H1-antihistamines, omalizumab
- DOI : 10.1684/ejd.2017.3085
- Page(s) : 455-63
- Année de parution : 2017
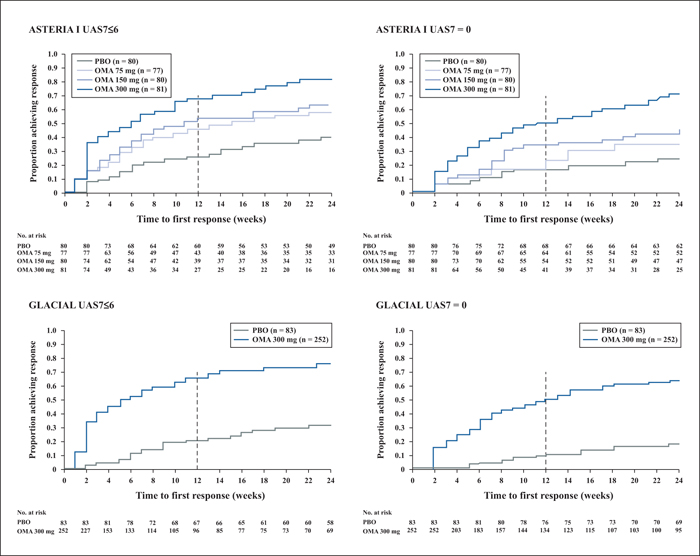
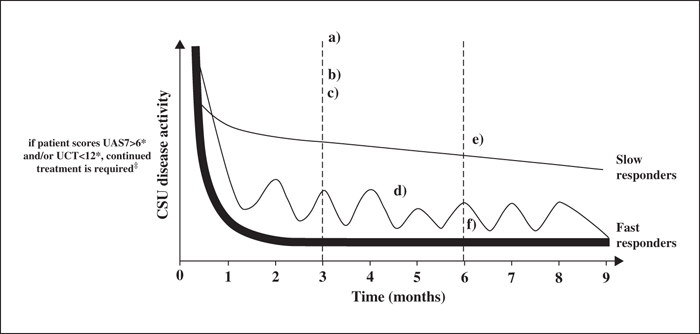
Omalizumab (a recombinant, humanized anti-immunoglobulin-E antibody) has been shown in three pivotal Phase III trials (ASTERIA I, II and GLACIAL) and real-world studies to be effective and well-tolerated for the treatment of chronic spontaneous urticaria (CSU), and is the only licensed third-line treatment for CSU. However, the definition of response to omalizumab treatment often differs between clinical trials, real-world studies, and daily practice of individual physicians globally. As such, a consensus definition of “complete”, “partial” and “non-response” to omalizumab is required in order to harmonize treatment management and compare data. Here, it is proposed that a disease measurement tool, for example, the 7-Day Urticaria Activity Score (UAS7) or Urticaria Control Test (UCT) is required for defining response. The addition of quality of life measurements is helpful to gain insight into a patient's disease burden and its changes during treatment. A potential omalizumab treatment approach based on speed and pattern of response at 1-3 and 3-6 months is suggested. In cases where there is no response during the first 1-3 months, physicians should consider reassessing the original CSU diagnosis. Moreover, in patients showing partial response at 12 weeks, treatment with omalizumab should be continued in order to maximize the possibility of achieving symptom control. If patients have a UAS7>6 and/or UCT<12, then continued treatment is advised, dependent on physician judgement and patient expectations. In treatment responders, omalizumab treatment can be resumed at a later stage after discontinuation with the same degree of symptom control.