Magnesium Research
MENUThe art of magnesium transport Volume 28, numéro 3, July-August-September 2015
Illustrations
- Mots-clés : distal convoluted tubule, kidney, TRPM6, CNNM2, SLC41A3, PCBD1
- DOI : 10.1684/mrh.2015.0388
- Page(s) : 85-91
- Année de parution : 2015
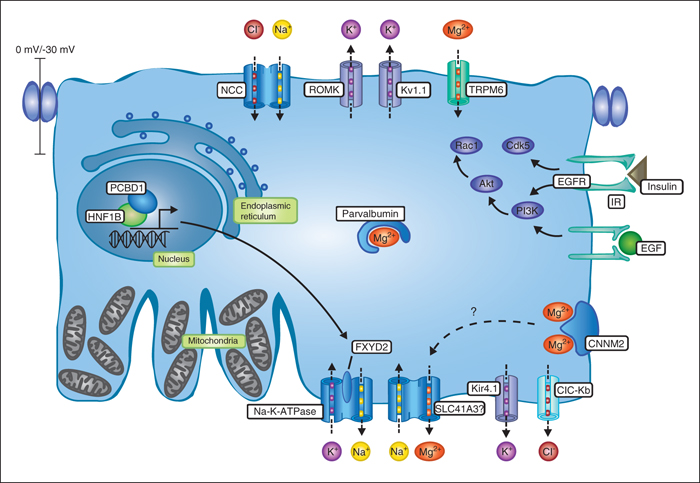
Patients with hypomagnesemia suffer from a wide range of symptoms including muscle cramps, cardiac arrhythmias and epilepsy. Disturbances in body Mg2+ homeostasis can often be attributed to increased Mg2+ excretion by the kidney. Within the kidney, the distal convoluted tubule (DCT) segment determines the final Mg2+ excretion, since no reabsorption takes place beyond this segment of the nephron. On 21st of January 2015, Jeroen de Baaij defended his thesis “The Distal Convoluted Tubule: the Art of Magnesium Transport”, in which he aimed to identify new genes involved in Mg2+ reabsorption in the DCT. This review summarizes the main findings of his graduate research. TRPM6 mediates apical Mg2+ entry into the DCT cell and is highly regulated by EGF, insulin and pH. ATP and flavagline compounds have been characterized as new regulators of TRPM6 activity, providing novel pathways to target Mg2+ disturbances. Using isolated primary DCT cells from mice, PCBD1 was identified as a new transcriptional regulator of Mg2+ transport in the DCT. Indeed, patients with PCBD1 mutations were shown to suffer hypomagnesemia and MODY5-like diabetes. Subsequently, the work presented in the thesis focused on the elucidation of the basolateral Mg2+ extrusion of the DCT cell. In vivo studies using SLC41A3-knockout mice suggest that SLC41A3 may act as Mg2+ extrusion mechanism. CNNM2 has long been hypothesized to transport Mg2+ at the basolateral membrane of the DCT. However, by determining the protein topology and homology modeling of the CBS domains, it was argued that CNNM2 is rather an Mg2+-sensing mechanism. Follow-up studies using 25Mg2+ isotopes showed that CNNM2 increases Mg2+ uptake when overexpressed in HEK293 cells. Additionally, by knocking down cnnm2 in zebrafish, CNNM2 was demonstrated to be essential for brain development and Mg2+ homeostasis. Mutations in CNNM2 were shown to cause hypomagnesemia, seizures and intellectual disability. Altogether, this thesis established the importance of Mg2+ reabsorption in the DCT to health and disease. Combined, continued efforts of clinicians, geneticists, and researchers are necessary to improve the care of hypomagnesemic patients and increase our understanding of Mg2+ reabsorption in the DCT.