European Journal of Dermatology
MENUThe safety and efficacy of four different fixed combination regimens of adapalene 0.1%/benzoyl peroxide 2.5% gel for the treatment of acne vulgaris: results from a randomised controlled study Volume 28, numéro 4, July-August 2018
- Mots-clés : acne, adapalene/benzoyl peroxide, emollient lotion, treatment regimen, moisturizer, management of treatment side effects
- DOI : 10.1684/ejd.2018.3367
- Page(s) : 502-8
- Année de parution : 2018
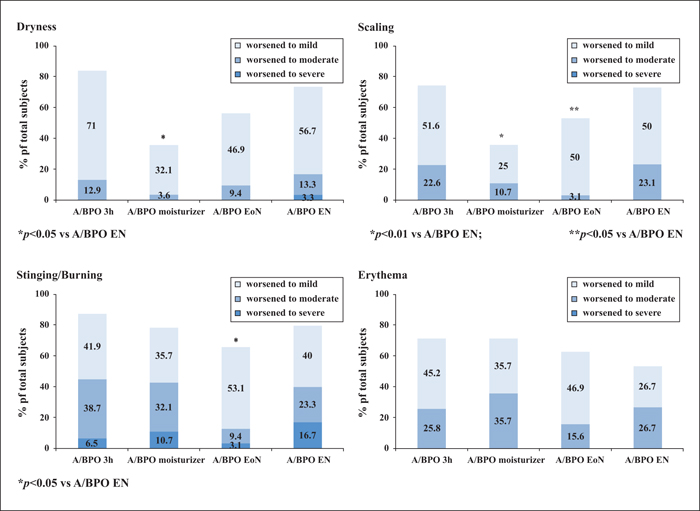
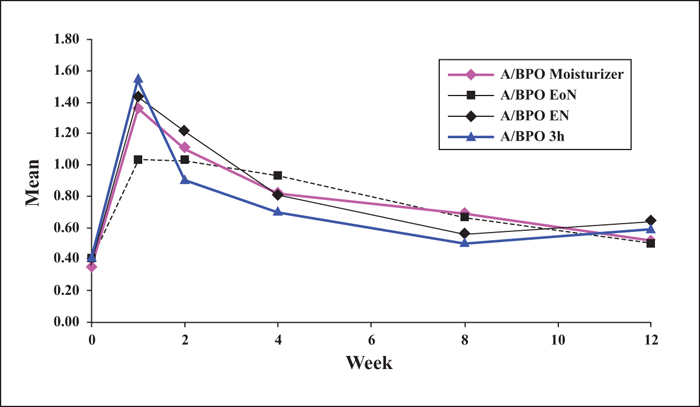
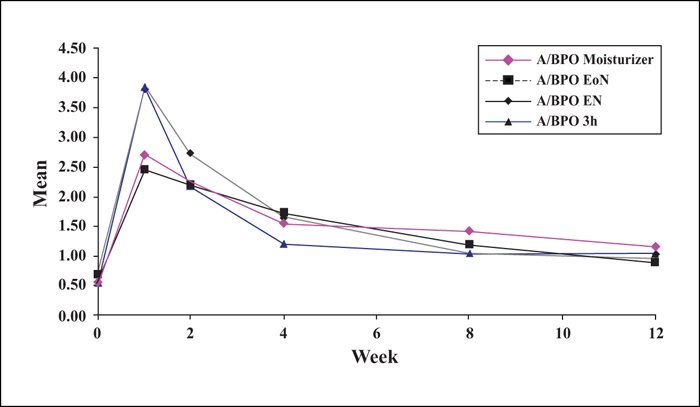
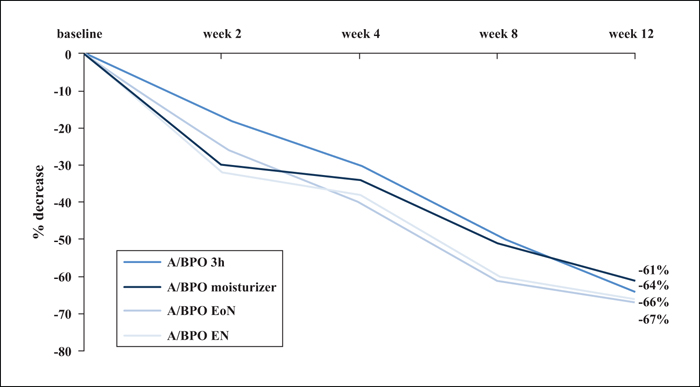
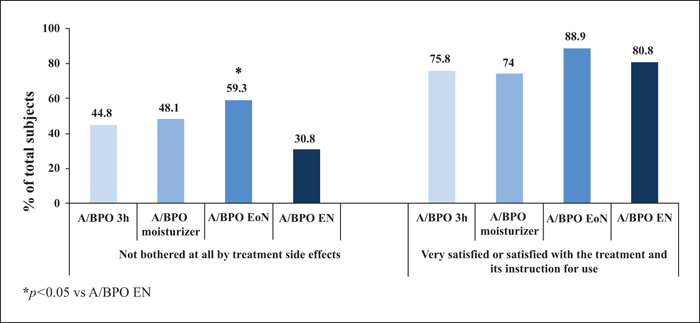
Background: Combined use of a retinoid and antimicrobial is recommended for acne, however, local tolerability issues may compromise patient adherence and treatment outcome. Objectives: This multicentre, single-blinded controlled study was designed to determine whether modified adapalene/benzoyl peroxide (A/BPO, Epiduo®, Galderma, France) regimens improve local tolerability during the first four weeks of treatment without impairing efficacy at Week 12. Materials & Methods: In total, 120 subjects with mild-to-moderate acne received, during the first four weeks, A/BPO daily overnight (A/BPO-EN), A/BPO daily for three hours (A/BPO-3h), A/BPO daily overnight and a provided moisturizer lotion (A/BPO-moisturizer), or A/BPO every other night (A/BPO-EoN). Local tolerance assessments included signs and symptoms, global worst score (GWS), and total sum score (TSS). Efficacy was assessed based on lesion counts, investigator global assessment (IGA), and total lesion count reduction. Adherence, subject satisfaction, and overall safety were also assessed. Results: The mean TSS was significantly reduced at Week 1 with A/BPO-EoN vs. A/BPO-EN (p<0.05), and A/BPO-EoN led to the lowest GWS and a decrease in severity of stinging/burning and erythema (p<0.05). The A/BPO-moisturizer regimen prevented dryness and scaling compared with the A/BPO-EN regimen. The median decrease in lesions from baseline was similar in all groups: up to 67% for total, 72% for inflammatory, and 70% for non-inflammatory lesion counts. Adherence, IGA, patient satisfaction, and overall safety were excellent. Conclusions: Modulating treatment regimens during the first four weeks improved local tolerability without impacting overall efficacy outcome after 12 weeks and may improve treatment adherence during the first weeks of therapy.