European Journal of Dermatology
MENUMarjolin ulcers after cultured epidermal autograft in severely burned patients: a rare case series and literature review Volume 31, numéro 6, November-December 2021
- Mots-clés : cultured epidermal autograft, massive burns, graft site malignancy, Marjolin ulcer, verrucous squamous cell carcinoma
- DOI : 10.1684/ejd.2021.4174
- Page(s) : 759-70
- Année de parution : 2021
Background
The advent of tissue engineering and the clinical applications with cultured epidermal autograft (CEA) have improved the prognosis of severely burned patients. Marjolin ulcers (MUs) are a well-known complication of burns. These malignant neoplasm transformations of burn scars are usually squamous cell carcinomas with a higher incidence of regional metastases. Radical surgery remains the treatment of choice.
Objectives
To identify cases of malignant transformation occurring at sites of CEA in a cohort of 68 massively burned patients.
Materials & Methods
A retrospective single-centre study was performed from April 2017 to June 2019 at the Military Hospital of Clamart (France). A total of 34 patients treated between 1991 and 2013 (including one post-mortem) were included.
Results
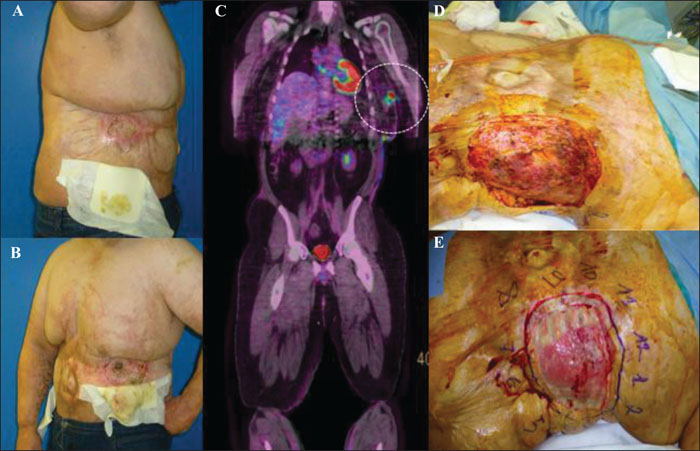
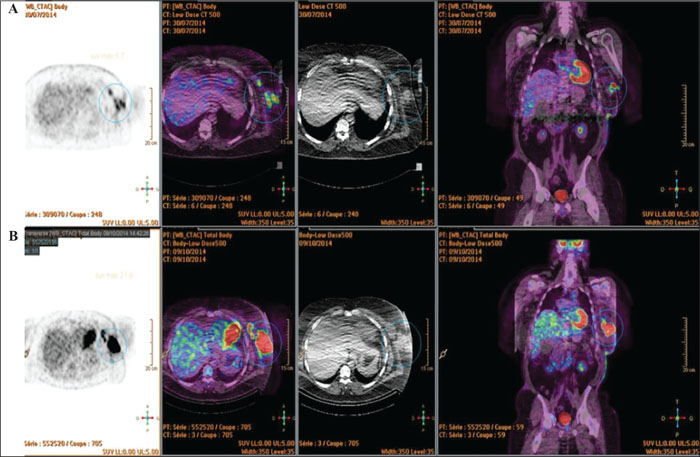
Four cases of squamous cell carcinoma occurred in areas previously covered by CEA. The data from clinical and histopathological examination as well as treatment modalities are presented. One patient died as a result of the evolution of his squamous cell carcinoma, and two others required salvage amputation due to locoregional recurrence. The prevalence of these CEA-MUs is estimated at between 5.9% and 11.7% and the person-time incidence rate of CEA-related MUs is about 5.9 per 1,000 persons-years. In our study, the average time to malignant transformation seems considerably shortened (32-35 years for “classic burn MU” versus 15.7 years for CEA-MU).
Conclusion
This first documented case series of CEA-MUs demonstrates the need to inform caregivers and educate patients. Further studies are needed to specify the true incidence of CEA-graft site malignancy.