European Journal of Dermatology
MENUReal-life, long-term data on efficacy, safety, response and discontinuation patterns of omalizumab in a Greek population with chronic spontaneous urticaria Volume 30, numéro 6, November-December 2020
- Mots-clés : chronic spontaneous urticaria, urticaria, omalizumab, angioedema, allergy, IgE inhibitors
- DOI : 10.1684/ejd.2020.3919
- Page(s) : 716-22
- Année de parution : 2020
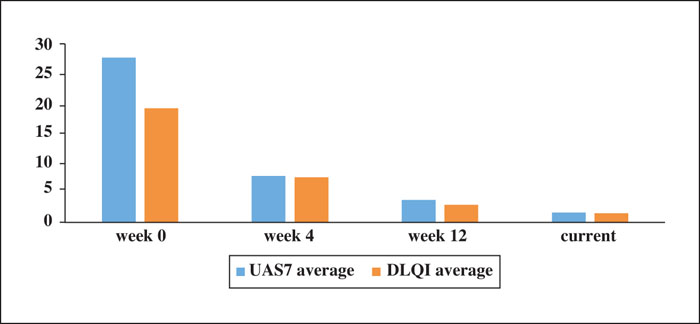
Background: Omalizumab is approved for the treatment of chronic spontaneous urticaria (CSU) not responding to antihistamines. Data on omalizumab use in real-world settings and in different populations are lacking. Objectives: To record our five-year experience of omalizumab use in patients with refractory CSU in a real-world setting. Materials & Methods: A retrospective analysis of medical records of 80 patients with refractory CSU was performed. Demographic, and clinical characteristics, patterns of response, discontinuation strategies and rate of recurrence were analysed. Results: Eighty individuals were included. UAS7 and DLQI significantly decreased from baseline. Complete response was achieved in 86.3%. Late response was observed at 27.5% of the patients. After discontinuation, 21.7% of patients reinitiated omalizumab due to relapse. The mean number of omalizumab administrations up to first discontinuation was 6.8 (based on an approach to shorten the treatment interval). Only 15.0% of patients experienced adverse events during treatment. Conclusion: Omalizumab, with long-term management, was highly effective and safe in achieving control of refractory CSU, with more favourable responses compared to Phase III clinical trials.