European Journal of Dermatology
MENUPsoriatic patients treated with secukinumab reach high levels of minimal disease activity: results from the SUPREME study Volume 31, numéro 5, September-October 2021
- Mots-clés : psoriasis, body surface area (BSA), Dermatology Life Quality Index (DLQI), Investigator Global Assessment (IGA), Minimal Disease Activity (MDA), Psoriasis Area and Severity Index (PASI), secukinumab
- DOI : 10.1684/ejd.2021.4150
- Page(s) : 630-7
- Année de parution : 2021
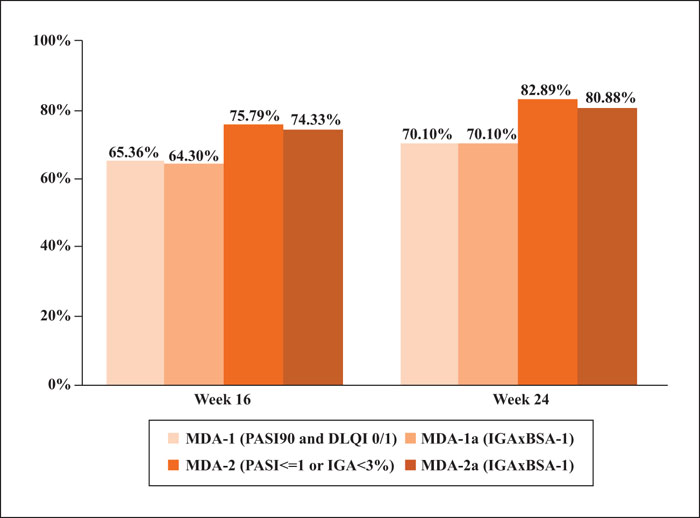
Background: Achieving minimal disease activity (MDA) represents an ambitious and sustainable therapeutic goal in psoriasis. Clear criteria for defining MDA in psoriasis are lacking. Objectives: The primary outcome was to evaluate the effect of 300 mg secukinumab in achieving MDA in patients with psoriasis and identify the most useful criteria to define MDA in such patients. The secondary outcome was to identify clinical factors influencing MDA. Materials & Methods: In this post hoc analysis of the SUPREME study, in which 433 patients were enrolled, MDA was assessed using established criteria: ≥90% improvement in Psoriasis Area and Severity Index (PASI 90) and Dermatology Life Quality Index 0/1 (MDA-1), PASI score ≤1 or body surface area (BSA) <3% (MDA-2), or Investigator Global Assessment x BSA (MDA-1a and MDA-2a), for which cut-off values were obtained in patients achieving MDA-1 and MDA-2, respectively. Results: After 16 weeks of secukinumab, 65% and 76% of the evaluable population achieved MDA-1 and MDA-2, respectively; at Week 24, this was 70% and 83%. Factors that positively influenced MDA at Week 16 were younger age, lower weight and body mass index, absence of depression and anxiety, and lower serum levels of complement C3 and high-sensitivity C-reactive protein. MDA-1a and MDA-2a were achieved by 64% and 74% of patients at Week 16 and by 70% and 81% at Week 24, respectively. Conclusion: Patients treated with secukinumab achieved high levels of MDA at Weeks 16 and 24, regardless of the method used to calculate MDA.