Epileptic Disorders
MENUEnteral lorazepam is a promising weaning strategy for midazolam-responsive febrile infection-related epilepsy syndrome (FIRES): a case series Volume 24, issue 4, August 2022
- Key words: pharmacological coma, midazolam-dependent, enteral, lorazepam, FIRES
- DOI : 10.1684/epd.2022.1438
- Page(s) : 667-76
- Published in: 2022
Objective
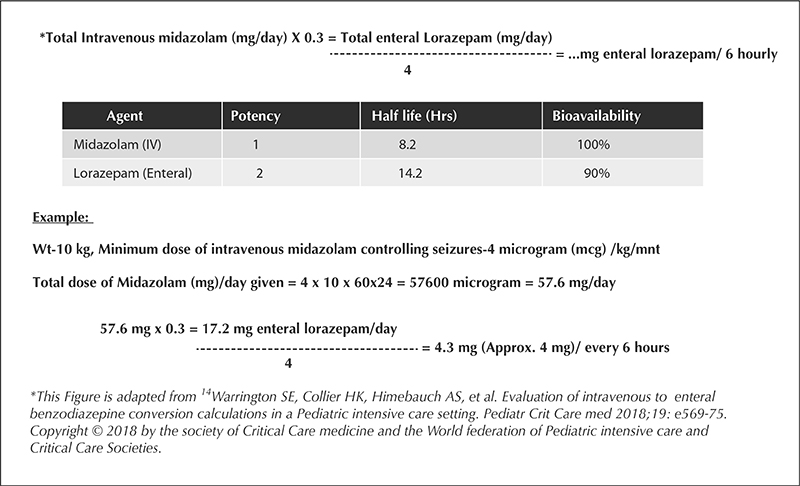
Prolonged and repetitive cycles of pharmacological coma, with midazolam or other general anaesthetics, is often the mainstay for seizure control in febrile infection-related epilepsy syndrome (FIRES). Here we present our experience of enteral lorazepam as an effective weaning substitute in midazolam-dependent patients.
Methods
This was a retrospective study based on a review of medical records of all FIRES patients who had received enteral lorazepam as a weaning substitute for midazolam, between January 2020 and July 2021. The patients were divided into an early group (lorazepam initiated after one failed attempt to wean off midazolam) and late group (lorazepam initiated after two or more failed attempts). The conversion from intravenous midazolam to enteral lorazepam was also calculated, and epilepsy outcome at follow-up was also assessed.
Results
Seven patients (five males) were eligible. The median age at onset of FIRES was seven years (range: 4-14). A median of six (range: 6-8) anti-seizure medications (ASMs) had failed (including clobazam in two and clonazepam in one) to control seizures. The early and late lorazepam groups were comparable regarding the maximum midazolam dose for seizure control, total ASMs tried and days to wean off midazolam. The median (range) duration of hospital stay was 27 days (22-46) in the early group, compared to 51 days (40-78) in the late group. The early group patients were also on fewer ASMs (median: 3; range: 3-5) compared to the late group (median: 5; range: 4-6) at discharge. Five patients were sedated with initial lorazepam dose, but this side effect resolved on dosage reduction. On follow-up, all seven patients had seizure recurrence. In four, seizures recurred on reducing lorazepam, however, in three of these patients, this was resolved by escalating the dose.
Significance
Enteral lorazepam can be an effective weaning substitute for midazolam-dependent children with FIRES. Early introduction of enteral lorazepam was associated with reduced duration of hospital stay.