European Journal of Dermatology
MENUMicrowave therapy for cutaneous human papilloma virus infection Volume 27, numéro 5, September-October 2017
Cutaneous HPV infection is common and warts are thought to affect most people at some time during their lives. Point prevalence estimates range from 0.8% to 4.7% of the population and two million people seek medical advice about warts each year in the UK [1], yet treatment options are poor and a meta-analysis has shown no significant benefit over placebo [2]. Although skin is most frequently infected by “non-oncogenic” HPV, most HPV-associated skin squamous cell carcinomas are diagnosed in persistent and recalcitrant verrucae and the majority contain HPV16 [3].
HPV infects the basal epithelial cells of cutaneous and mucosal keratinised epithelia and infection is mainly controlled by T cell-mediated immunity [4]. HPV-specific CD8+ lymphocytes are critical for clearance of HPV viral warts [4] and individuals treated with immunosuppression to prevent organ graft rejection do not clear HPV infections. In healthy individuals, induction of HPV-specific CD8+ T cells with topical imiquimod (TLR7 agonist) has been shown to facilitate wart clearance [5, 6]. However, tissue penetration is a limiting factor for the therapeutic potential of imiquimod on most non-mucosal sites.
Other modalities of thermal ablation have previously been investigated for the treatment of warts [7-10]. Direct heat ablation is now rarely used because of scarring and subsequent morbidity. The most widely used physical modality is liquid nitrogen application (cryotherapy) to the skin [11]. This causes tissue destruction and in a recent meta-analysis of randomised controlled trials, this therapy has been shown to have low efficacy in the management of common warts (with a mean clearance on all sites of 49%) [12]. Microwaves (30 MHz to 30 GHz) exist in the electromagnetic spectrum between radiofrequency and visible light and have been widely used as a means for delivering heat energy to induce thermal ablation in the treatment of cancer, especially for inoperable liver tumours [13], but have not been previously applied to skin. Recent technological advances have enabled development of a hand-held device to deliver targeted application of microwave therapy to skin. We set out to test the potential of this new modality as a treatment for warts in a Phase 1, open-label, uncontrolled clinical study. It was observed in the first few cases that the warts shrank and resolved without obvious necrosis, tissue damage, or inflammation. Hence, we hypothesised that somehow anti-HPV immunity was being activated. We therefore undertook morphological and histological analysis of microwave-treated human skin and investigated for evidence of enhanced anti-HPV immunity. We demonstrated that, even at low energy levels, microwave therapy potentiates cutaneous immunity to HPV.
Methods and materials
Patients and in vivo microwave treatment
The study was approved by the local research ethics committee in accordance with the declaration of Helsinki. Individuals with treatment-refractory plantar warts were recruited. The diagnosis of plantar wart was confirmed by a podiatrist experienced in management of such lesions. A clinically significant wart was defined as >one year duration, with at least two previous failed treatments (salicylic acid, laser, cryotherapy, needling, and surgical excision). Exclusions were pregnancy or breast feeding, pacemaker in situ, metal implants within the foot or ankle, co-morbidities affecting immune function, or capacity to heal. At each study visit, a complete examination of the affected area was undertaken and a quantitative measure of pain and neuromuscular function assessed. No dressing was required and volunteers continued normal everyday activities after treatment with no restrictions.
A total of 32 volunteers with 54 foot warts were enrolled into the study (17 males and 15 females; age range: 22-71 years; mean: 44·79 years [SD: 13.019]). Sixteen were solitary and 38 multiple-type warts (e.g. mosaic verrucae). Mean lesion duration was 60.54 months (range: 12-252) and diameter 7·43 mm (range: 2-38 mm; SD: 6.021). At the conclusion of the study period, one patient had been lost to follow-up and two patients had withdrawn (n = 3; four warts) but were retained in the statistical analysis, classified as unresolved lesions.
Microwave treatment (Swift®, Emblation Medical Ltd., UK) of the most prominent plantar wart was titrated up, as tolerated to 50 J over a 7-mm diameter application area (130 J/cm2) over 5 seconds (10 watts for 5 seconds). Lesions >7 mm received multiple applications until the entire surface of the wart had been treated. If the wart persisted, treatment was repeated at one week, one month, three months, and 12 months. Response to treatment was assessed by the same investigator as binary; “resolved” or “unresolved”. Resolution was indicated by fulfilling three criteria: (1) lesion no longer visible; (2) return of dermatoglyphics to the affected area; and (3) no pain on lateral compression. Pain was assessed using a 10-point visual analogue scale.
Human skin and blood samples
Skin and blood samples for microwave experiments were acquired from healthy individuals as approved by the local Research Ethics Committee in adherence to Helsinki Guidelines.
Histological analysis
Skin samples were treated immediately ex-vivo with microwaves (Swift s800; Emblation Ltd., UK) or liquid nitrogen therapy and punch biopsies taken from treated skin were sent for histological analysis or placed in culture media.
Histological analysis of hematoxylin and eosin (H&E)-stained tissue sections was undertaken following fixation and embedding in paraffin wax. DNA damage was assessed by staining for single-stranded and double-stranded DNA breaks by TUNEL assay using the ApopTag® In Situ Apoptosis Detection Kit (Millipore, UK). Following culture, supernatants were collected and analysed for lactate dehydrogenase (LDH) release using the Cytotoxicity Detection Kit (Roche applied science) as a measure of apoptosis.
Cell culture and in vitro microwave treatment
Primary keratinocytes were obtained from pooled neonatal foreskin donors (Lonza, Switzerland) and cultured in keratinocyte growth medium 2 (PromoCell) at 37˚C, 5% CO2, until 70-90% confluency for use in experimental work (P4-P10).
Human skin explant cultures and human HaCaT keratinocytes were cultured in calcium-free DMEM (ThermoFisher Scientific) with 100 U/mL penicillin, 100 μg/mL streptomycin, 1 mM sodium pyruvate, 10% foetal bovine serum (FBS), and supplemented with calcium chloride at 70 μM final concentration.
Microwave treatment of cells in culture was delivered in a flat-bottomed well using the Swift device applied directly to the plastic base from the underside. To assess whether the plastic caused loss of microwave energy in our system, the 150 J Swift programme applied through the culture well base delivered a temperature rise of 18.6̊C (SD: 1.1) to 200 g of culture media, equivalent to ∼15.61 J (SD: 0.92). Thus, it could be estimated that 15 J applied ex vivo would be equivalent to ∼150 J as tested here in vivo. However, energy loss during skin application would reduce this difference, but calculation of the precise transfer of energy to skin in vivo was not possible, so we estimate that the dose delivered in vitro is up to 10-fold lower than that by direct skin application ex vivo. To avoid confusion, the setting on the Swift system is the energy level referred to throughout the manuscript (in human and in vitro studies).
Lymphocytes were cultured in RPMI-1640 medium with 100 U/mL penicillin, 100 μg/mL streptomycin, 1 mM sodium pyruvate, and 292 μg/mL L-glutamine, supplemented with 10% FBS or 10% heat-inactivated human serum (HS). HaCaT cells were cultured to sub-confluency to avoid cell differentiation and used in assays at passage 60-70. Cells were plated at 2.5 × 103 cells/well in 96-well flat plates (Corning Costar) and cultured overnight to reach confluence. HaCaTs were washed once with PBS before treatment with microwaves, liquid nitrogen (10 seconds), or with LPS+IFN-γ (1 ng/mL+1,000 U/mL). Cells were cultured for 24 hours before supernatants were harvested. HPV16 E7 protein was expressed in E. coli at the Protein Core Facility of Cancer Sciences Unit, University of Southampton. Endotoxin was removed using Detoxi-Gel endotoxin removal using columns (Thermo Scientific).
For HPV-specific T cell lines, PBMCs were isolated from HLA-A2-positive individuals, as previously described [14]. PBMCs were seeded at 2-4 × 106 cells/well in 24-well culture plates and 10 μg/mL ninemer HLA-A2-restricted HPV16 epitope LLM (LLMGTLGIV) [15] was added; cells were cultured in 1 mL RPMI+10% HS. On Day 3, cells were fed with RPMI+10% HS+IL-2 (200 IU/mL), and then fed again on Day 7 or when needed. After Day 10, HPV-specific T cells were harvested for cryopreservation before testing against HPV using ELISpot assays.
To generate monocyte-derived dendritic cells (moDCs), CD14+ cells, were positively isolated from PBMCs by magnetic separation using CD14 microbeads (Milentyi Biotec, UK), according to the manufacturer's protocol. Cells were washed and resuspended in RPMI+10% FBS+250 U/mL IL-4 and 500 U/mL GM-CSF. At Day 3, cells were fed with RPMI+10% FBS+IL-4 and GM-CSF, and then harvested on Day 5 for use in functional assays.
ELISpot, flow cytometry and qPCR
Keratinocytes (HaCaTs or primary as indicated) were treated with microwaves at various energy settings before removal of supernatant at various time points. MoDCs were treated overnight with keratinocyte supernatant, then washed twice before incubation with LLM peptide (10 μg/mL for 2 hours) or HPVE7 protein (10 μg/mL for 4 hours) before a further wash. Human IFN-γ ELISpot (Mabtech, Sweden) was undertaken, as per the manufacturer's protocol and as reported previously [14]. moDCs at 1 × 103 were plated with autologous HPV peptide-specific T cells at a ratio of 1:25. Spot forming units (sfu) were enumerated with ELISpot 3.5 reader (AID, Germany).
MoDCs were treated with HaCaT supernatant and harvested at 24 hours for flow cytometric analysis of cell phenotype. Cells were stained with violet LIVE/DEAD stain (Invitrogen, ThermoFisher, UK) for 30 minutes at 4̊C, then washed with PBS+1% BSA and stained with antibodies PerCP-Cy5.5 anti-HLA-DR, FITC anti-CD80, FITC anti-CD86, or PE anti-CD40 (Becton Dickinson, UK) for 45 minutes at 4̊C. Cells were washed, then resuspended in PBS+1% BSA, and analysed using the BD FACSAria and the FlowJo v10.0.08 analysis software.
The expression of chosen genes was validated with quantitative PCR using the TaqMan gene expression assays for target genes: YWHAZ (HS03044281_g1), IRF1 (Hs00971960_m1), and IRF4 (Hs01056533_m1) (Applied Biosystems, Life Technologies, UK) in human skin, and treated as indicated. RNA extraction (RNeasy mini kit, Qiagen) and reverse transcription (High-Capacity cDNA Reverse Transcription Kit, Applied Biosystems; ThermoFisher Scientific UK)) were carried out accordingly to the manufacturer's protocol.
Results
Treatment of human papilloma virus infection in humans with microwave therapy
Of the 32 volunteers with severe warts, 54 treatment-refractory plantar warts were treated with microwave therapy (figure 1A, B). At the end of the study period, of the 54 warts treated, 41 had resolved (75.9%) and nine remained unresolved (16.7%), and two patients (with three warts [5.6%]) withdrew from the study and one patient (with one wart [1.9%]) was lost to follow-up. The mean number of days to resolution was 79.49 days (SD: 34.561; 15-151 days). Of the resolving lesions, 94% had cleared after three treatments (figure 1C). No significant difference in resolution rates between males and females (p = 0·693) was observed. Statistically significant reductions in pain were observed as treatment progressed (p<0.0001) (figure 1D). Adverse events were minimal. One patient reported transient pain from the treatment which required a simple oral analgesic (paracetamol) and resolved within 24 hours. This individual withdrew from the study. No further adverse events were reported. No cases of scarring were recorded following completion of treatment. No cases of neuromuscular dysfunction were reported.
Microwave treatment of human skin
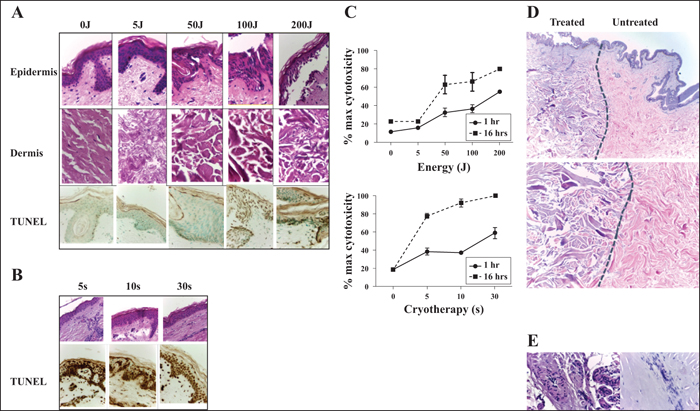
Human skin has not previously been reported to be treated with microwave therapy, therefore, we proceeded to undertake a full histological analysis of treated skin. Skin removed during routine surgery was treated ex vivo and one hour after treatment punch biopsies were taken and fixed for histological processing. Neither macroscopic nor histological changes were noted with the lowest energy setting (5 J). At 50 J, mild macroscopic epidermal changes only were noted, and microscopically minor architectural changes and slight elongation of keratinocytes were seen without evidence of altered dermal collagen. At higher energies (100/200 J), gross tissue contraction was visible macroscopically. Microscopic changes in the epidermis were prominent, showing spindled keratinocytes with linear nuclear architectural changes and subepidermal clefting (figure 2A). Dermal changes were prominent at energies of 100 J and above and showed a homogenous hyalinised zone of papillary dermal collagen, thickened collagenous substances, and accentuation of basophilic tinctorial staining of the dermal collagen with necrotic features (figure 2A). These features are similar to electro-cautery artefacts and suggest that at >100 J, there is the potential to coagulate proteins and induce scarring. Histological analysis both at 16 hours and 45 hours showed similar changes (data not shown).
In clinical practice, cryotherapy is delivered to the skin by cryospray, which is time-regulated by the operator. In contrast to microwave therapy, minimal epidermal or dermal architectural change was identified with cryotherapy at standard treatment duration times (5-30 seconds), but did show a dose-dependent clumping of red blood cells in vessels (figure 2B).
Tissue release of LDH acts as a biomarker for cellular cytotoxicity and cytolysis. To examine the extent of cell death induced by microwave irradiation, human skin was treated with 0, 50, 100 or 200 J before punch excision of the treated area and incubation in medium for one hour or 16 hours. Measurement of LDH revealed a dose-dependent induction of tissue cytotoxicity with increasing microwave energies (figure 2C). In line with the lack of histological evidence of cellular damage, at 5 J, cytotoxicity of microwave application was equivalent to control. Early cytotoxicity was not prominent at 50 J, but became more evident after 16 hours. Higher energy levels induced more prominent cytotoxic damage. In contrast to microwave therapy, liquid nitrogen treatment of skin induced cytotoxicity at the lowest dose both at one hour and 16 hours.
Terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL) identifies cells in the late stage of apoptosis. Analysis at 0, 5, 50, 100 and 200 J identified increased cellular apoptosis in the epidermis above 100 J (figure 2A). In contrast, cryotherapy induced significant epidermal and dermal DNA fragmentation (figure 2B).
The physics of microwave therapy suggests a sharp boundary between treated and untreated tissue with minimal spreading of the treated field. This was borne out histologically by a clear demarcation between treated areas extending vertically from the epidermis through the dermis (figure 2D). Examination of the dermis showed that microwave therapy modified skin adnexae, inducing linear nuclear architectural changes in glandular apparatus, micro-thrombi, fragmented fibroblasts, and endothelial cells (figure 2E).
Microwave induction of immune responses in skin
We first examined the response of keratinocytes to microwave therapy in vitro. In analysing in vitro the effects of microwave therapy, it was necessary to apply the microwave treatment through culture dish plastic. Thus, the energy setting in vitro is equivalent to a lower energy setting than with direct application in vivo (see above). In keratinocyte monolayers (HaCaT), apoptosis was induced by microwave therapies above 100 J in vitro (figure 3A). Only above the apoptotic threshold (100 J) were surface phenotypic changes of cellular activation noted in viable cells with increased expression of HLA-DR, CD40, and CD80 (figure 3B). Next, we utilised a model of skin cross-talk of keratinocyte signalling to dermal dendritic cells. Initially, we observed strong activation of MoDCs primed with supernatant from microwave-treated keratinocytes (data not shown), but we wished to disentangle the pro-inflammatory effects of apoptosing/necrotic cells from viable cell cross-talk. Therefore, keratinocytes were treated with microwave therapy as above, and washed after eight hours to remove dead or apoptotic cells. Treated keratinocytes were then incubated for a further 16 hours before supernatant collection to prime moDCs, which had not been directly exposed to microwave therapy. The supernatants induced potent induction of moDC activation with increased expression of CD86, CD80, and to a lesser extent, CD40 (figure 3C).
We next set out to model the functional outcome on skin dendritic cells following microwave treatment of keratinocytes. Keratinocyte monolayers (HaCaT) were untreated, or microwave- or cryotherapy treated before supernatant harvesting. Supernatant-primed DCs were pulsed with a nine-amino acid HLA-A2 epitope (LLM) from human papilloma virus (HPV) E7 protein and cultured with an autologous HPV-specific CD8+ T cell line. As expected, in all conditions, the moDCs efficiently presented HPV peptide to HPV-specific CD8+ T cells, inducing IFNγ (figure 4A). However, dendritic cell presentation of HPV is dependent upon cross-presentation to the MHC class I pathway. Therefore, we also tested the capability of untreated, microwave-treated or cryotherapy-treated KC-primed moDCs to present HPV E7 protein to an HLA-matched HPV-specific CD8+ T cell line. Strikingly, only microwave-treated KCs were capable of priming moDCs to enhance cross-presentation (figure 4B). To explore the potential mechanism of keratinocyte response to microwave therapy, we confirmed up-regulation of HSP-70 in response to microwave therapy of keratinocytes (figure 4C). Although, the assay used did not distinguish constitutive from inducible HSP-70, we clearly demonstrated global increase in HSP-70 expression following microwave therapy. Additionally, IL-6, but not IL-1β or TNF-α, was expressed in response to microwave stimulation, which suggests that alternative inflammatory signalling pathways from that seen in cryotherapy-treated cells are induced by microwave stimulation (figure 4D). To further explore the potential innate immune signalling pathways in keratinocytes following microwave therapy, we examined IRF1 and IRF4. These transcription factors are key regulators of dendritic cell activation of adaptive immunity. We show that microwave therapy induced down-regulation of IRF1 and up-regulation of IRF4 (figure 4E).
Discussion
This is the first study to investigate the potential efficacy of locally delivered microwaves in the treatment of cutaneous viral warts. In this uncontrolled pilot study, we report a complete resolution rate of 75.9% of recalcitrant plantar warts (with an average lesion duration of over five years). This compares very well with previous reports of plantar wart resolution for salicylic acid and or cryotherapy (23-33%) [16].
For all novel therapies, adverse events are critical but we did not identify a strong signal for adverse events. As with current physical treatments for warts, discomfort is expected for the patient. During the study, patients generally reported that for a typical five-second treatment, they endured moderate discomfort for approximately two seconds, which immediately diminished after the treatment had completed. In addition, it was commonly noted that discomfort was less with subsequent treatments. One male patient withdrew from the study after one treatment, citing the pain of treatment as the reason. In the study design phase, pre-operative use of topical anaesthetic cream was tested, but appeared to do little to mitigate the pain (unpublished data) and it was felt that the pain of local anaesthetic injection would exceed that normally experienced during a microwave treatment. Following microwave therapy, patients did not require dressings or special advice as no wound or ulcer was caused, allowing the patient to continue normal activity. The short microwave treatment time (five seconds) offers a significant clinical advantage over current wart therapies, such as cryotherapy and electro-surgery. Within five seconds, microwaves penetrate to a depth of over 3.5 mm at the energy levels adopted for the study [17]; possibly a greater depth than can be attained by cryosurgery or laser energy devices. Moreover, microwaves, like all forms of electro-magnetic radiation, travel in straight lines and energy is deposited in alignment with the “beam” emitted from the device tip with little lateral spread, meaning minimal damage to surrounding tissue, as confirmed in this study. Microwaves induce dielectric heating. When water, a polar molecule, is exposed to microwave energy, the molecule is excited and rotates to align with the alternating electro-magnetic field. At microwave frequencies, the molecule is unable to align fully with the continuously shifting field resulting in heat generation. Within tissues, this acts to rapidly elevate temperatures. This process increases cellular temperature because it does not depend on tissue conduction. Microwave treatment produces no vapour or smoke unlike ablative lasers and electro-surgery, eliminating the need for air extraction systems due to the risk of spreading viral particles within the plume [18].
Although microwave therapy has been considered a tissue ablation tool, we observed minimal skin damage after treatment with 50 J, yet good clinical responses were seen. Therefore, we investigated whether there was evidence to support an induction of anti-HPV immunity by microwave therapy. The critical nature of CD8+ T cell immunity for host defence against HPV skin infection is well established and supported by the observation of increased prevalence of infection in immunosuppressed organ-transplant recipients [19], and that induction of protection from HPV vaccines is mediated by CD8+ T cells [20]. We show here that microwave therapy of skin induces keratinocyte activation and cell death through apoptosis. However, in vitro microwave-primed keratinocytes are capable of signalling to dendritic cells and enhancing cross-presentation of HPV antigens to CD8+ lymphocytes at microwave energy levels equivalent to or lower than that used in the clinical study, which offers a potential explanation for the observed response rate in our clinical study. In vitro evidence suggests that this is likely to be mediated by cross-talk between microwave-treated skin keratinocytes and dendritic cells, through induction of danger-associated molecular patterns (DAMPs), such as HSP-70 in keratinocytes, resulting in up-regulation of DC CD40 and CD80/86 and subsequent enhanced cross-presentation of HPV proteins to CD8+ T cells. Microwave therapy also specifically induced enhanced IL-6 synthesis from keratinocytes. IL-6, is a pro-inflammatory mediator, important in anti-viral immunity, which has been recently shown to induce rapid effector function in CD8+ cells [21]. Thus, IL-6 up-regulation may provide an important additional mechanism for microwave-induced anti-viral immunity. The intriguing contrast between cryotherapy and microwave therapy revealed a far greater release of IL-1β and TNF-α with cryotherapy which, in addition to the lesser IL-6 induction, may offer potential to utilise the treatments for different situations where IL-1β/TNF-α-driven inflammation may be preferable, or vice versa. Additionally, the specificity of inflammatory pathways induced by each modality may explain why cryotherapy and microwave stimulation may not show equal effectiveness in the same disease.
IRFs have been shown to be central to the regulation of immune responses [22-24]. IRF4 is essential for differentiation of cytotoxic CD8+ T cells [25, 26], but up-regulation in dendritic cells has also been shown to enhance CD4+ differentiation [23], therefore, this pathway may potentially enhance both CD8+ immunity and T-cell help following microwave treatment. IRF1 expression has been previously reported to be modulated by HPV infection, but different models have shown opposite outcomes [27, 28]. We show down-regulation of IRF1 in human skin in association with a microwave therapy, which supports the proposal of IRF-1 as a therapeutic target in HPV infection [28].
This study is the first of its kind to study microwaves in the treatment of plantar warts in vivo. Further work to examine the immune infiltrate in microwave-treated warts is planned. Whilst we acknowledge the limitations of the uncontrolled, non-randomised design, the promising results shown here suggest that a randomised controlled study with a larger sample size is warranted to confirm the efficacy of this treatment.
Disclosure
Financial support: All financial support was provided by an investigator-led research grant from Emblation Medical Ltd., the maker of the Swift microwave system used in this study. Emblation Medical Ltd. had no input into the design, data capture, analysis, or manuscript preparation. After completion of the work and following the first draft of the manuscript, I.B. has become a consultant for Emblation Medical Ltd. Conflict of interest: none.